[A0003]
Attila Kiss
a, József Jekő b , Albert Lévai a , Tamás Patonay a
a Department of Organic Chemistry,
Lajos Kossuth University, H-4010 Debrecen P.O.B. 20, Hungary b
ICN-Hungary, H-4440 Tiszavasvári, Hungary
E-mail: [email protected]
Received: 1 August 1999 / Uploaded: 16 August 1999
a
-Hydroxy ketones including their cyclic derivatives are important synthetic building blocks. Chiral, non-racemic a-hydroxy ketones (and esters) are widespread in natural products. Therefore, there is a high demand to develop efficient methods for the construction of their enantiopure or at least enantiomerically enriched representatives and a number of procedures have been published in the last decades [1,2]. Our interest in the synthesis of a-hydroxy benzo(hetera)cyclanones, particularly 3-hydroxychromanones, prompted us to investigate the synthetic utility of the recently developed electrophilic chiral oxidizing system, namely dimethyldioxirane (DMD)/Jacobsen's catalyst [3] in the oxyfunctionalization of enol derivatives. Enol acetates were choosen as prochiral substrates because their higher stability as compared to silyl enol ethers and their high reactivity toward DMD [4].Scheme 1
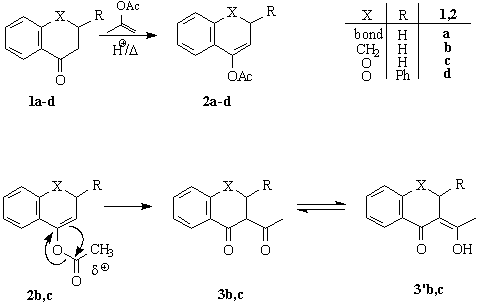
When enol acetates 2a,b were reacted with DMD under the standard conditions, only a-hydroxy ketones 5a,b and a-acetoxy ketones 6a,b have been isolated, traces of the intermediate epoxides 4a,b could only be detected by TLC. When the reaction was conducted in the presence of anhydrous K2CO3 as additive to eliminate of any water and trace of acids, we were able to obtain epoxide 4b in pure, crystalline form but analogous reaction of 2a afforded only a mixture of 4a,5a and 6a. Oxidation of heterocyclic enol acetates 2c,d proceeded smoothly and furnished only the corresponding a-ketols 5c,d and acetate 6c,d without any detectable amount of epoxides 4c,d (Scheme 2). We can conclude that the stability of epoxides 4 highly depends on the structure of the substrate. Some selected results of oxidation experiments are shown in Table 1.
Scheme 2
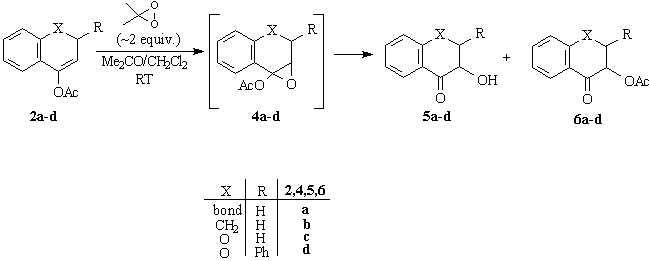
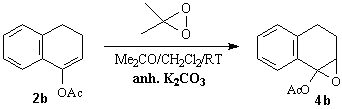 Table 1. Oxidation of enol
acetates 2 by DMD
Table 1. Oxidation of enol
acetates 2 by DMD
Substrate |
Additive |
T (oC) |
NMR product ratio |
Isolated yields (%) 4 |
Isolated yields %) 5 |
Isolated yields (%) 6 |
2a |
none |
RT |
- |
0 |
31 |
49 |
2a |
anh. K2CO3 |
-20 |
4a:5a:6a=75:2:23 |
- |
- |
- |
2b |
none |
RT |
5b:6b=35:65 |
0 |
29 |
63 |
2b |
10 % H2O | RT |
5b:6b=46:54 |
0 |
24 |
22 |
2b |
anh. K2CO3 | -20 |
- |
84 |
traces |
traces |
2c |
anh. K2CO3 | -20 |
5c:6c=43:57 |
0 |
15 |
34 |
2d |
anh. K2CO3 | -20 |
- |
0 |
34a,c |
17b,c |
a
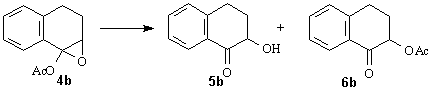
pure 2,3-trans diastereomer b mixture of diastereomers; 2,3-cis/2,3-trans=63:37 (1H NMR)Availability of the moderately stable, crystalline epoxide 4b allowed us to investigate the ring-opening process of a-acetoxy epoxides. The slow reaction in the absence of acids even at elevated temperature (Table 2, Entries 1, 2) clearly indicate that acetates 6a-d do not form in the well-documented thermal rearrangement [2g, 6] but in a different, probably acid-catalysed pathway. 2-Acetoxytetralone (6b) proved to be stable under the rearrangement conditions, this control experiment unequivocally indicates that 5b and 6b form in two distinct way. Appropriate conditions were also found to transform epoxide 4b into a-ketol 5b as a major or exclusive product (Table 2, Entries 4,5) (Scheme 3). Scheme 3
 Table
2. Ring-opening of 1-acetoxy-1,2-epoxytetralin (4b)
Table
2. Ring-opening of 1-acetoxy-1,2-epoxytetralin (4b)
Entry |
Conditions |
Conversion (%) |
NMR product ratio (5b:6b) |
Isolated yields (%) 5b |
|
1 |
abs. PhH/RT/2d |
no reaction |
no reaction |
no reaction |
no reaction |
2 |
abs. PhH/RT/2d |
13 |
58:42 |
- |
- |
3 |
SiO2/CH2Cl2/RT 2d |
100 |
48:52 |
39 |
|
4 |
TFA (1 equiv.) MeOH/RT/1h |
100 |
91:9 |
85 |
9 |
5 |
K2CO3/MeOH /N2/RT/20 min |
100 |
100:0 |
86 |
0 |
Oxidation of enol acetate 2b by DMD in the presence of R,R- or S,S- Jacobsen's salen catalyst afforded optically active intermediate epoxides 4b wich were transformed by SiO2 (Kieselgel) treatment into the corresponding chiral, non-racemic 2-hydroxy- (5b) and 2-acetoxytetralone (6b) (Scheme 4).
Scheme 4
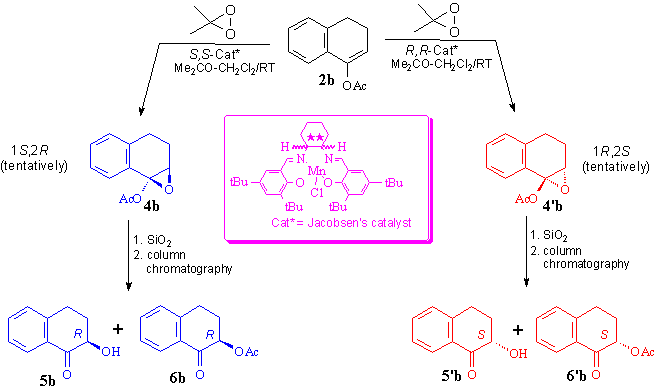
Absolute configurations of the products were determined by comparison of the sign of the measured specific rotation with literature values [2a,d]. The effect of the catalyst loading on the optical purity of the intermediate epoxide 4b (Figure 1) and ring-opened products 5b, 6b (Figure 2) as well as the effect of used co-ligands (axial ligands) on the optical purity of products 4b, 5b and 6b (Figure 3) and on the e.e. values [7] of products 5b, 6b (Figure 4) were also studied. Best enantioselectivities were achieved by using 10 mol% Jacobsen's catalyst in the presence of 40 mol% imidazole or 4-phenylpyridine-N-oxide (PPNO) as co-ligand. Figure 1

Figure 2
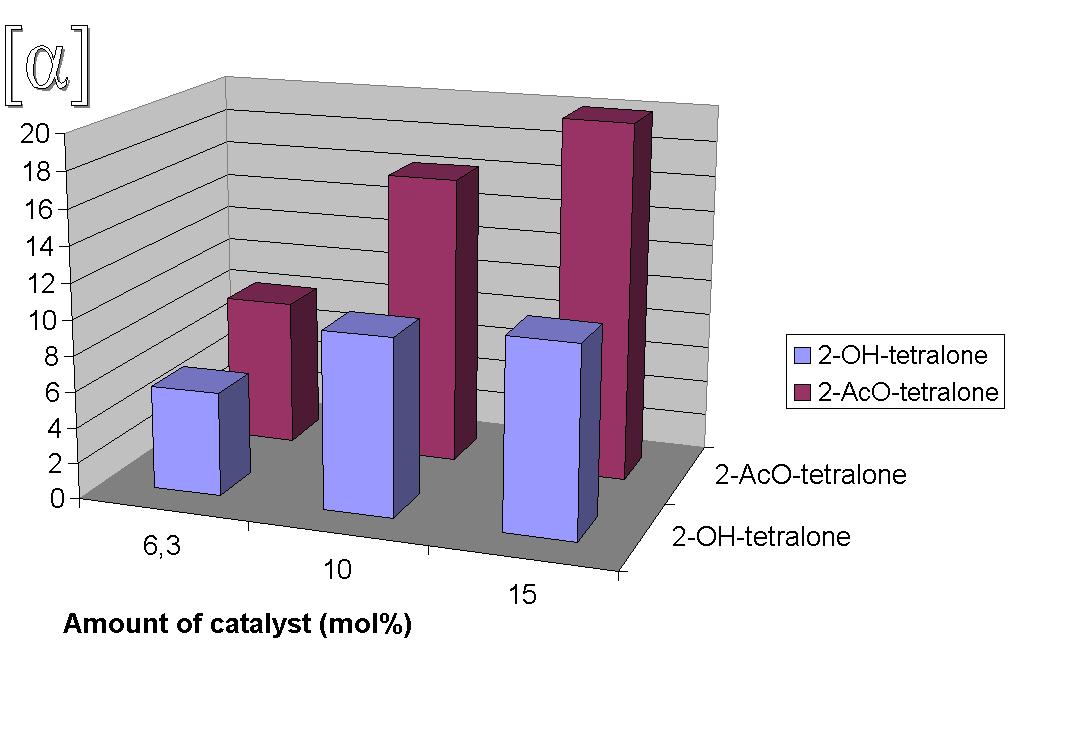 Figure 3
Figure 3
 Figure 4
Figure 4 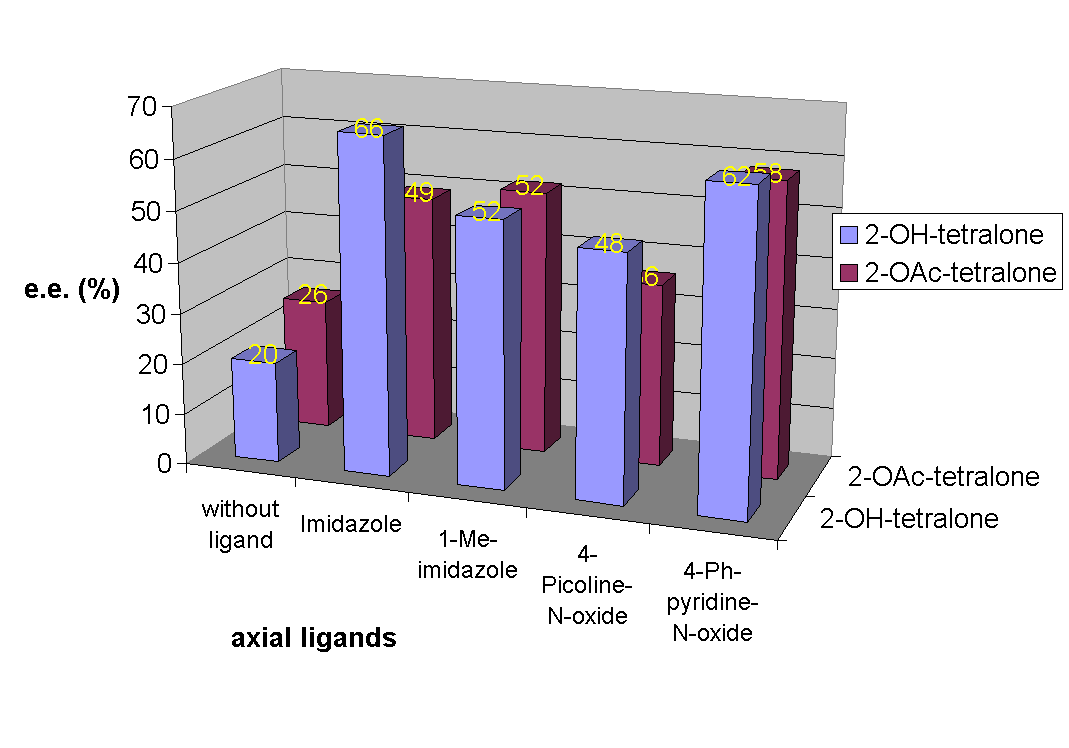
In summary, enol acetates of benzo(hetera)cyclanones are synthetically useful substrates for dioxirane oxidation. The use of Jacobsen's catalyst as chiral oxidant in the presence of DMD as oxygen donor allows enantioselective a-oxyfunctionalisation. Enhancement of enantioselectivity (up to 60-70% e.e.) could be achieved by using various co-ligands.
Acknowledgements: Financial support of National Fund of Science and Research (OTKA # T22290) is highly appreciated. References
[1] Davis, F.A.; Clark,
C.K.; Kumar, A.; Chen, B-C. Chem.Rev. 1992, 92, 919.
[2] [a] Davis, F.A.; Clark,
C.K.; Kumar, A.; Chen, B-C. J.Org.Chem. 1994, 59,
1184; [b] Adam, W.;
Prechtl, F. Chem.Ber. 1994, 127, 667; [c] Adam, W.; Fell,
R.T.; Hoch, U.;
Saha-Möller, C.R.; Schreiner, P. Tetrahedron:Asymmetry1995, 6,
1047; [d] Adam, W.;
Diaz, M.T.; Fell, R.T.; Saha-Möller, C.R.
Tetrahedron:Asymmetry 1996,
7, 2207; [e] Adam, W.; Fell, R.T.; Saha-Möller,
C.R.; Zhao, C.G. Tetrahedron:Asymmetry
1998, 9, 397; [f] Adam, W.; Fell, R.T.;
Stegmann, V.R.;
Saha-Möller, C.R. J.American Chem. Soc. 1998, 120, 708;
[g] Zhu, Y.; Tu, Y.;
Yu, H.; Shi, Y. Tetrahedron Lett. 1998, 39, 7819 and the
references cited
therein.
[3] [a] Adam, W.; Jekő, J.;
Lévai, A.; Nemes, Cs.; Patonay, T.; Sebők, P.Tetrahedron
Lett. 1995, 36,
3669; [b] Adam, W.; Jekő, J.; Lévai, A.; Majer, Zs.; Nemes, Cs.;
Patonay, T.;
Párkányi, L.; Sebők, P. Tetrahedron:Asymmetry 1996, 7, 2437;
[c] Adam, W.; Fell,
R.T.; Lévai, A.; Patonay, T.; Peters, K.; Simon, A.; Tóth, G.
Tetrahedron: Asymmetry
1998, 9, 1121 [d] Lévai, A.; Adam, W.; Fell, R.T.;
Gessner, R.; Patonay,
T.; Simon, A.; Tóth, G. Tetrahedron, 1998, 54, 13105.
[4] Adam, W.; Hadjiarapoglou,
L.; Jäger, V.; Klicić, J.; Seidel, B.; Wang, X.
Chem.Ber., 1991,
124, 2361.
[5] [a] Saito, R.; Izumi, T.;
Kasahara, T. Bull. Chem. Soc. Japan, 1973, 46,1776;
[b] Climent,
M.J.; Garcia, H.; Iborra, S.; Miranda, M.A.; Primo, J. Tetrahedron
1991, 47, 9289;
[c] Patonay, T.; Hoffman, R.V. J.Org.Chem.1994, 59, 2902.
[6] [a] McDonald , Rearrangements
of a-Halo Epoxides and Related
a-Substituted Epoxides in Mechanism of Molecular Migrations, Vol.3.;
Thyagarijan, B.S., Ed.;
Wiley, 1971; 67-107; [b] Catsoulacos, P.
J.Heterocycl.Chem.
1986, 23, 321.
[7] Determined by HPLC (chiral
column Chiralcel OB, hexane-iPrOH=9:1).
All comments on this poster should be sent by e-mail to (mailto:[email protected] ona.edu)
[email protected] with A0003 as the message subject of your e-mail.