[a00038]
Department of Organic Chemistry, State Academy of Fine Chemical Technology, Vernadsky Avenue 86, Moscow 117571, Russia. Phone/Fax (095) 431-6332.
Abstract: A new general two-steps synthesis of 5-functionalized 4-hydroxyhexahydropyrimidine-2-thiones/ones has been developed. This synthesis are based on preparation of the a-azido and a-tosyl(thio)ureas 2, 5 followed by reaction with enolates of a-functionally substituted ketones. All the obtained hydroxypyrimidines 8a-c are readily converted into the corresponding 5-functionalized 1,2,3,4-tetrahydrohydropyrimidine-2-thiones/ones 9a-c by heating in the presence of acids. Treatment of the 4-acylsubstituted 4-hydroxyhexahydropyrimidine-2-thiones/ones 8b with bases in acetonitrile gives the N-acyl-N'-(b-oxoalkyl)thioureas/ureas 10 in result of rearrangement including C(4)-C(5)-bond cleavage in 8b. Reaction of the (thio)ureides 10 and the 5-acylsubstituted 4-hydroxyhexahydropyrimidine-2-thiones/ones 8b with KOH in water leads to the 4-hydroxyhexahydropyrimidine-2-thiones/ones 11 without the acyl group at the C(5) position. Treatment of the ethyl 4-hydroxy-2-thioxohexahydropyrimidine-5-carboxylates 8a with bases in acetonitrile depending on the starting compounds structure yields either the 5,6-dihydro-2-thiouracils 12, 13 or the products of C(4)-C(5)-bond cleavage 14.
Keywords: N-(azidomethyl)thiourea, N-(1-tosyl-1-alkyl)thioureas/ureas, 4-hydroxyhexahydropyrimidine-2-thiones/ones, 1,2,3,4-tetrahydropyrimidine-2-thiones/ones, N-acyl-N'-(b-oxoalkyl)thioureas/ureas, 5,6-dihydro-2-thiouracils.
![]() Introduction
Introduction
![]() Results and Discussion
Results and Discussion
![]() Synthesis of
a-azido and a-tosyl substituted
thioureas and ureas
Synthesis of
a-azido and a-tosyl substituted
thioureas and ureas
![]() Synthesis of
5-functionalized 4-hydroxyhehaxydropyrimidine-2-thiones/ones
Synthesis of
5-functionalized 4-hydroxyhehaxydropyrimidine-2-thiones/ones
![]() Synthesis of
5-functionalized 1,2,3,4-tetrahydropyrimidine-2-thiones/ones
Synthesis of
5-functionalized 1,2,3,4-tetrahydropyrimidine-2-thiones/ones
![]() Rearrangement
of 5-acyl-4-hydroxyhexahydropyrimidine-2-thiones/ones in the presence of bases
Rearrangement
of 5-acyl-4-hydroxyhexahydropyrimidine-2-thiones/ones in the presence of bases
![]() Synthesis
of 4-hydroxyhexahydropyrimidine-2-thiones/ones
Synthesis
of 4-hydroxyhexahydropyrimidine-2-thiones/ones
![]() Transformations
of ethyl 4-hydroxy-2-thioxohexahydropyrimidine-5-carboxylates in the presence of bases
Transformations
of ethyl 4-hydroxy-2-thioxohexahydropyrimidine-5-carboxylates in the presence of bases
![]() Conclusion
Conclusion
![]() References
References

4-Hydroxyhexahydropyrimidine-2-thiones/ones are of great interest due to their high synthetic potential. It can be explained by presence of the interconnected N,O-acetal and thiourea/urea moieties in their molecules as well as by ability of their conversion to the acyclic isomeric forms (ring-chain isomerism) [1]. Thus, it is not surprising that these compounds are versatile precursors in syntheses of a large variety of heterocycles. For example, they were used in syntheses of fully and partly hydrogenated pyrimidines [2], 1,3-thiazines [3], pyridines [4], condensed heterocycles [5], etc. Besides, 4-hydroxyhexahydropyrimidine-2-thiones/ones manifest various useful properties. Some of these compounds possess herbicidal [6] and radioprotective [7] activities, improve quality of textiles [8], etc.
At present time there are three main methods for the synthesis of 4-hydroxyhexahydropyrimidine-2-thiones/ones from acyclic precursors:
It should be noted that according to these procedures it is difficult or impossible to
obtain 4-hydroxyhexahydropyrimidine-2-thiones/ones bearing a functional group at the C(5)
position of pyrimidine ring. That is why 5-functionalized analogs of
4-hydroxyhexahydropyrimidine-2-thiones/ones are scarcely known, and their chemistry and
biological properties remain unexplored. We report here on a novel, general method for
convenient preparation of these heterocycles via ureidoalkylation and describe some their
transformations.
In our retrosynthetic analysis of 5-functionalized 4-hydroxyhexahydropyrimidine-2-thiones/ones we considered the reaction of a-substituted (thio)ureas (A) with enolates of carbonyl compounds (B).
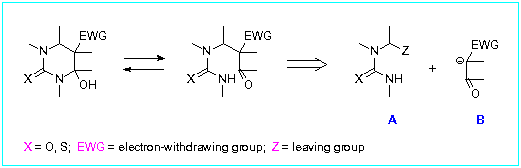
Effectiveness of the target pyrimidines synthesis depends on proper choice of leaving group Z in ureidoalkylation reagent (A). Recently we have demonstrated [12] that readily available cyclic (thio)ureas bearing azido or arylsulfonyl groups at the a-position to nitrogen are efficient in ureidoalkylation. We proposed that (thio)ureas A possessing the same leaving groups could serve as key intermediates in the synthesis of 4-hydroxyhexahydropyrimidine-2-thiones/ones.
a
-Azido or a-tosyl substituted (thio)ureas 2, 5 served as the starting compounds for the pyrimidine synthesis. They were readily obtained in 1-2 steps from thiourea or urea in good yields. Thus, N-(azidomethyl)thiourea 2 and N-(tosylmethyl)thiourea 5 (R = H) were prepared by the reaction of methylolthiourea 1 with hydrazoic acid or p-toluenesulfinic acid in water (Scheme 1). a-Substituted tosyl(thio)ureas 5 were synthesized by the treatment of thiourea or urea with aliphatic or aromatic aldehydes and p-toluenesulfinic acid in water. The obtained (thio)ureas 2, 5 owing to their good purity were used for the pyrimidine synthesis without further purification.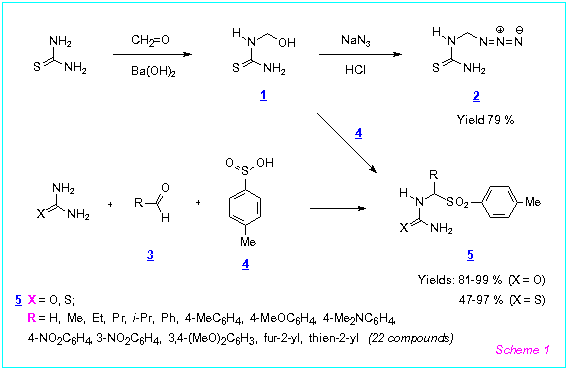
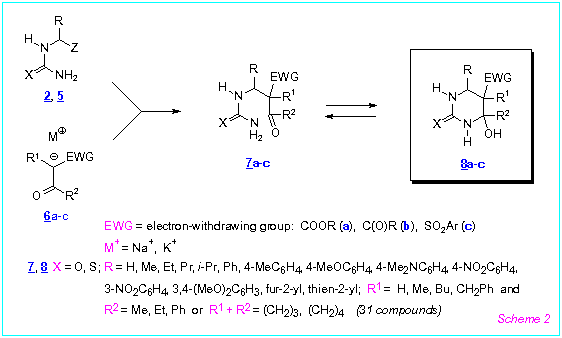
We found that the (thio)ureas 2, 5 reacted redily (r.t., 2-6 h) with
enolates of various b-oxoesters, 1,3-dicarbonyl compounds or a-arylsylfonylketones 6a-c generated in situ by
treatment of the corresponding CH-acids with KOH in ethanol or with NaH in acetonitrile to
give the corresponding 5-functionalized 4-hydroxyhexahydropyrimidine-2-thiones/ones 8a-c
in a regioselective manner (Scheme 2). The products were formed in high
diastereomeric purity (de 52-100 %).
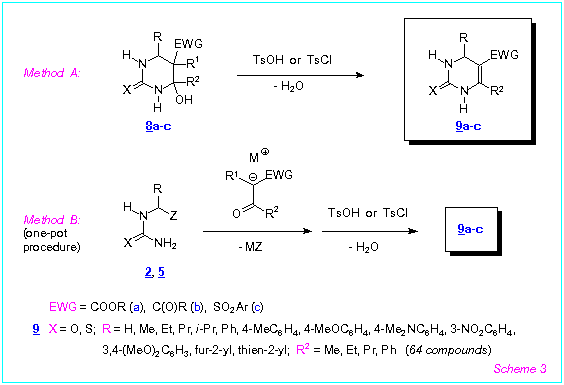
We investigated behaviour of the compounds 8a-c towards acids and bases. We
found that these compounds were readily dehydrated by refluxing in ethanol or acetonitrile
in the presence of TsOH or TsCl (method A) to afford 5-functionalized
1,2,3,4-tetrahydropyrimidine-2-thiones 9a-c in excellent yields (Scheme 3). The
compounds 9a-c were also prepared in good overall yields according to one-pot
procedure by reaction of the (thio)ureas 2, 5 with the enolates 6a-c
followed by acidification and refluxing of the reaction mixtures (method B).
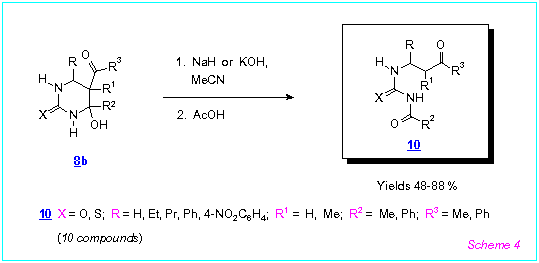
We demonstrated that 5-acyl-4-hydroxyhexahydropyrimidine-2-thiones/ones 8b
underwent rearrangement in the presence of NaH or NaOH in acetonitrile to produce, after
acidification of the reaction mixtures, unknown N-acyl-N'-(b-oxoalkyl)thioureas
and ureas 10 (Scheme 4).
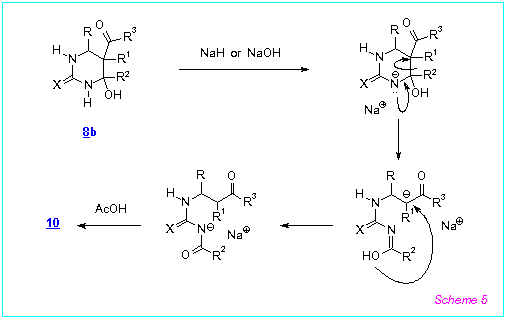
Proposed mechanism of this unusual rearrangement is presented on Scheme 5 and
includes C(4)-C(5)-bond cleavage in the anion of 8b.
The obtained (thio)ureides 10 were easily hydrolized by treatment of KOH in
water to give the 4-hydroxypyrimidines 11 (Scheme 6). The latter were also
produced directly starting from the 5-acyl-4-hydroxypyrimidines 8b (KOH, water).
Probably, this transformation proceeds via the retro-Claisen reaction in the
acyclic isomeric form of 8b.

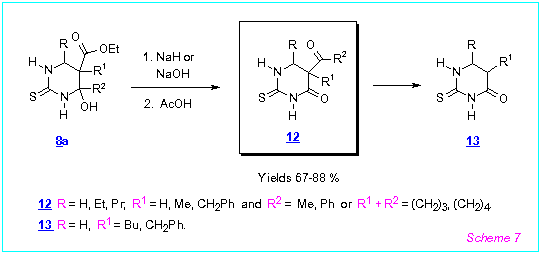
Reaction of ethers of 4-hydroxy-2-thioxohexahydropyrimidine-5-carboxylic acids 8a
with bases (NaH or NaOH in acetonitrile) is more complex than for 8b. In the case
of 6-unsubstituted or 6-alkyl substituted 8a, their transformation into
5-acyl-5,6-dihydro-2-thiouracils 12 took place (Scheme 7). Apparently, this
reaction proceeds via the acyclic isomeric form of 8a. Sometimes the
deacylation products of 12 namely 5,6-dihydro-2-thiouracils 13 were formed.
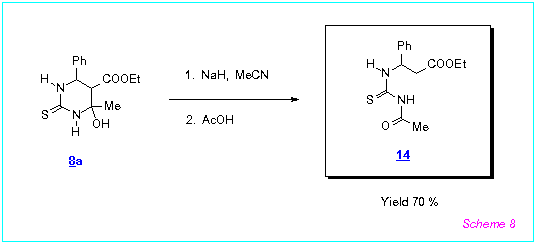
In the case of 6-phenyl substituted 8a, its rearrangement into the thioureide 14
occured (Scheme 8). Probably, mechanism of this rearrangement is the same as for
the synthesis of 10.
Thus, the present work demonstrates that 5-functionalized
4-hydroxyhexahydropyrimidine-2-thiones/ones can be prepared by reaction of readily
available a-tosyl or a-azido
substituted (thio)ureas with enolates of a-substututed ketones
(C-N-C-N + C-C ring construction). The obtained hydroxypyrimidine can serve as starting
compounds in syntheses of a large number of multifunctional pyrimidine-2-thiones/ones.
Mild reaction conditions, good overall yields, high flexibility make the described methods
of pyrimidine syntheses very promising.
1. Unkovskii, B.V.; Ignatova, L.A.; Zaitseva, M.G. Khim. Geterotsikl. Soedin. (Rus), 1969, 889; Unkovskii, B.V.; Ignatova, L.A.; Zaitseva, M.G.; Donskaya M.M. ibid, 1965, 586.
2. Shutalev, A.D.; Alekseeva, S.G. Khim. Geterotsikl. Soedin. (Rus), 1995, 377; Shutalev, A.D.; Komarova, E.N.; Pagaev, M.T.; Ignatova, L.A. ibid, 1993, 1259; Zigeuner, G.; Galatik, W.; Lintshinger, W.-B.; Wede, F. Monatsh. Chem., 1975, 106, 1219; Ovechkina, G.I.; Ignatova, L.A.; Ratomskaya, M.G.; Unkovskii, B.V. Khim. Geterotsikl. Soedin. (Rus), 1971, 1258; Zigeuner, G.; Rauter, W. Monatsh. Chem., 1965, 96, 1950.
3. Unkovskii, B.V.; Ignatova, L.A. Khim. Geterotsikl. Soedin. (Rus), 1969, 896.
4. Zigeuner, G.; Lintschinger, W.-B.; Fuchsgruber, A.; Kollmann, K. Monatsh. Chem., 1976, 107, 155.
5. Singh, H.; Kumar, S. Tetrahedron, 1987, 43, 2177; Zigeuner, G.; Kollmann, K.; Lintschinger, W.-B.; Fuchsgruber, A. Monatsh. Chem., 1976, 107, 183.
6. Krenzer, J. US Pat. 3951976 (1976), Cl C 07D 239/00.
7. Rolevitch, I.V.; Ignatova, L.A.; Shutalev, A.D. Unpublished data.
8. Petersen, H.; Pai, P.; Reichert, M. Eur. Pat. Appl. 33115 (1981), Cl D 06M 15/54.
9. Ignatova, L.A.; Shutalev, A.D.; Shingareeva, A.G.; Dymova, S.F.; Unkovskii, B.V. Khim. Geterotsikl. Soedin. (Rus), 1985, 260; Mathes, R.A. J. Amer. Chem. Soc., 1953, 75, 1747.
10. Shutalev, A.D.; Ignatova, L.A.; Unkovskii, B.V. Khim. Geterotsikl. Soedin. (Rus), 1984, 548; Ovechkin, P.L.; Ignatova, L.A.; Unkovskii, B.V. ibid, 1972, 941.
11. Zimmermann, R.; Brahler, B.; Hotze, H. Pat. 1065849 BRD (1961); Zimmermann, R.; Brahler, B.; Hotze, H. Angew. Chem., 1960, 72, 78.
12. Shutalev, A.D. Khim. Geterotsikl. Soedin. (Rus), 1993, 1645; Shutalev, A.D. ibid, 1993, 1389; Shutalev, A.D.; Ignatova, L.A. ibid, 1991, 228; Shutalev, A.D.; Ignatova, L.A.; Unkovskii, B.V. ibid, 1990, 133.
All comments on this poster should be sent by e-mail to (mailto:[email protected] ona.edu)
[email protected] with A0038 as the message subject of your e-mail.