[B0007]
Exploiting Poly(ethylene glycol) as a Matrixfor Liquid-Phase Organic Synthesis
Frank Sieber, Paul Wentworth Jr.* and Kim D. Janda*
Received: 11 August 1999 / Uploaded: 12 August 1999
Abstract
For many years solid polymers have been dominant both in parallel
syntheses and as supports for organic reagents.1,2
However, there are a number of major concerns associated with the use of insoluble
polymeric derivates under heterogeneous conditions such as lowered reactivities, side-side
interactions, extended reaction times and diffision-limited reactivity. The use of soluble
matrices3 such as poly(ethylene glycol)4
circumvents these problems while also allowing for routine monitoring of the reaction
progress. Additionally, the basis for developing soluble polymer-supported chemistry is
quite excellent since known solution-phase reaction conditions are preserved.5,6 Our efforts in this field have included the
development of soluble polymer-supported combinatorial libraries,7
catalysts,8 reagents,9-11
linker strategies12,13 and synthetic
methodology.14,15 This report details the
development and application of a poly(ethylene glycol) bound triarylphosphine reagent and
the optimization of a liquid-phase Stille cross-coupling reaction with subsequent
generation of a small library of biaryl, heterobiaryl and styryl derivatives in high
yields and purity.
Index
1. Development and Application of a
Poly(ethylene glycol)-Supported Triarylphosphine Reagent
1.1. Synthesis of Liquid-Phase
Phosphine Reagent 2
1.2. Liquid-Phase Ozonide Reduction
1.3. Liquid-Phase Wittig Reactions in
aqua
1.4. Regeneration of Spent Reagent 9
2. Poly(ethylene glycol) as a Soluble Polymer
Matrix for the Stille Cross Coupling Reaction
Our group recently communicated an extension o f PEG-supported chemistry into the field of functionalized polymers as reagent supports.16-18 We prepared a novel triarylphosphine derivative of PEG (1) and showed that, when compared to a heterogeneous commercially available counterpart, it possesses favorable reaction kinetics in both the Staudinger and Mitsunobu etherification reactions. The present study expands on those preliminary investigations.
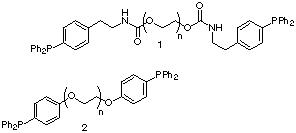
1.1. Synthesis of Liquid-Phase Phosphine Reagent 2. A concern with the original reagent 1 was the carbamate ester moiety incorporated as a linker between the triarylphosphine and the PEG support. The known base and acid sensitivity of the urethane linkage, coupled with worries about its lability in the presence of Lewis acids and metallating reagents, linked any further exploitation of PEG-supported triarylphosphines to an immediate replacement of this carbamate ester group. An aryl-alkyl ether moiety was chosen as the replacement linker, the chemical stability of which is comparable to that of the poly(ethylene oxide) backbone, therefore reagent 2 became the functionalized polymer of choice.19

The synthetic strategy towards 2 involves an initial concise preparation of the key hydroxyphosphine 3,20 followed by its attachment to PEG via the dimesylate 4.21 p-Bromophenol 5 was protected as the TBDMS ether 6, that was then phosphinylated under standard conditions20 to give the triarylphosphine 7. The silyl ether was removed and the resultant hydroxyphosphine 3 was obtained in 87 % yield for the three steps. Mesylate 4 was obtained by heating (50 oC) a neat solution of PEG3400 8 in methanesulfonyl chloride. No base was required under these conditions, which had the benefit that the purification of 6 was simplified from its original preparation.21 Etherification between 4 and 3 was performed in rigorously degassed DMF. Phosphine 2 was then isolated by addition of the reaction mixture into degassed diethyl ether, followed by filtration and sequential washing of the polymeric precipitate with i-Pr alcohol (to remove salts) followed by diethyl ether to give reagent 2 in 92 % yield, based on the weight of polymer isolated.
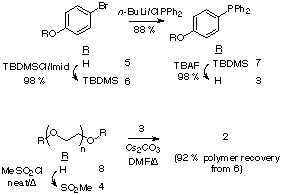
The oxidation state of the phosphine termini of 2 was routinely quantified by 31P NMR after certain periods of storage. This revealed that even after prolonged exposure to air (2-3 weeks) little oxidation to the phosphine oxide (< 5 %) occurs.The derivatization level of the polymer is also determined by inspection of the 1H NMR of 2. The chemical shift of a-methylene protons of PEG and its derivatives is a function of the moiety attached to the termini hydroxyl groups. For underivatized PEG 3400, the chemical shift of these a-protons is d 3.8 ppm, for PEG-mesylate 4, this changes to d 4.35 ppm (also detectable are the b-methylene protons d 3.74 ppm) and for the phosphine terminated polymer 2 this changes to d 4.16 ppm ( b-methylene protons d 3.64 ppm). Inspection of the 1H NMR of 2 shows that no mesylate 4 remains. Therefore there is high confidence that the polymeric phosphine 2 is quantitatively substituted with terminal triarylphosphine residues ( ca. 0.5 mmol g -1).
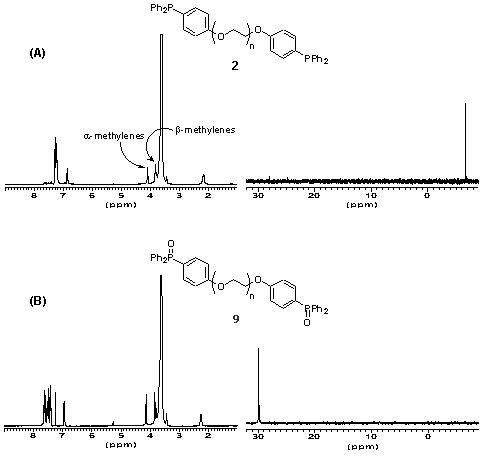
The ease of NMR-characterization of soluble polymer reagent 2 offers a considerable advantage over its solid-phase homologue which, in order to determine the oxidation state of the phosphine center, must involve either gel- or solid-phase NMR, or single-bead FTIR techniques.23
1.2. Liquid-Phase Ozonide Reduction. As an entry level into the utility of 2 in organic chemistry, the mild chemical decomposition of ozonides was selected. Ozonolysis of alkenes has a broad scope of application in organic chemistry, and the range of methods for destruction of the intermediary ozonides is equally broad. However, the two major methods of choice: Zn/acetic acid or dimethyl sulfide have limitations, a result of potential chemical substituent sensitivity in the former case, or noxious reagents and high-boiling point byproducts in the latter. For these reasons PPh3 is an excellent alternative.24 It has received only limited use in solution-phase chemistry however, because of the problems of removing both the excess PPh3 and reaction byproduct, triphenylphosphine oxide, from the reaction mixture. The use of polystyrene-supported PPh3 as a stoichiometric reagent for this reaction has been reported as an alternative and gives good to excellent yields of aldehydes.25 Therefore an important part of this study was a direct comparison between this new liquid-phase approach with 2, and a solid-phase approach with commercially available PPh3.A range of alkenes 10a-e were treated with ozone. The incipient ozonides were then decomposed into the product aldehydes ( 11a-e) by addition of one of three reagents: PPh3, polystyrene-supported PPh3, or PEG-supported PPh3 ( 2). The results are shown below (Table 1).
Table 1. Direct Comparison of Ozonide Hydrolysis Between Solution-Phase, Solid-Phase and Liquid-Phase Triphenylphosphine
entry |
alkene |
product |
PPh3b |
|
2 |
1 |
|
|
94 |
58 |
98 |
2 |
|
|
80 |
73 |
92 |
3 |
|
|
84d |
60d |
97d |
4 |
|
|
51 |
56 |
77 |
5 |
|
|
72 |
62 |
63 |
a
Reaction conditions: The respective triphenylphosphine (2eq.) was added to the ozonide (1eq.) of the alkene in CH2Cl2 and left to stir for 2 h. Yields based on HPLC comparison to authentic product standards. b Commercially available PPh3 (Aldrich Chem. Co.). c Commercially available polystyrene-PPh3 resin (Aldrich Chem. Co.). d Isolated yield.The isolation procedure for the PEG-supported reagent involved a simple precipitation of the spent reagent into ether, and concentration. This consistently removed > 99 % of the polymer byproduct. Passage of the ether concentrate through a short silica pad completely removed the remaining polymer traces, giving the product aldehydes in analytically pure form. The solid-phase reagent was separated from the reaction mixture by filtration. The product was isolated by then washing the resin with volumes of CH2Cl2. The solution-phase reactions were purified by silica gel chromatography to remove the excess PPh3 and the byproduct PPh3O.In all cases studied, the yields of aldehyde were highest with the soluble polymer-supported reagent 2 and with the exception of 11d the soluble reagent gave higher yields than the solid-phase reagent. This result may be a composite of three factors; increased reducing power of the soluble polymer-supported phosphine 2 when compared to both the solid- and solution-phase reagents, a result of the p-ethoxyether functionality which links the reagent to the soluble polymer support, the homogeneous nature of the ensuing chemical process when compared to the hetereogeneous resin reagent and the ease of isolation and purification of the products when compared with solution-phase strategy.
1.3. Liquid-Phase Wittig Reactions in aqua. Reagent 2 was subsequently transformed into the polymer-supported phosphonium salt 12 suitable for Wittig olefination in aqueous solution. Organic chemistry in water has been the focus of considerable efforts in recent years.26-28 The Wittig olefination reaction has also been much studied throughout the evolution of crosslinked polymer-supported reagents and considerable success has been achieved.29-34 Bernard and Ford34 noted an inverse correlation between the yield of alkene obtained from polymer-supported Wittig reagents with aldehydes and ketones in solution and the degree of divinylbenzene (DVB) cross-linking in the resin. Progressing from 0.5-20 % DVB crosslinking, i.e. from gel to macroporous resins, can result in a fall of 20 % in the yield of alkenes. These results suggested that a soluble polymer-supported phosphonium salt may give excellent yields in the Wittig reaction.The physical properties of the PEG backbone are such that a PEG-supported phosphonium salt would be eminently water soluble and offer the tantalizing prospect of facilitating the first soluble polymer-supported Wittig olefination reactions in aqua . Treatment of PEG-supported phosphine 2 with benzylbromide, followed by precipitation into diethyl ether yielded the soluble polymer-supported benzyl triarylphosphonium salt 12 in 81 % yield (based on the weight of polymer obtained). This polymeric phosphonium salt 12 is completely soluble in water at pH 7. The reaction of this novel Wittig salt (12) was then studied with a range of aldehydes ( 11a and 13a-d) in aqueous sodium hydroxide solution (Scheme 2, Table 2).
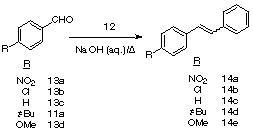
| NO2 | 13a | NO2 | 14a |
| Cl | 13b | Cl | 14b |
| H | 13c | H | 14c |
| t-Bu | 11a | t-Bu | 14d |
| OMe | 13d | OMe | 14e |
Table 2. Aqueous Wittig Reactions Phosphonium Salt 12 and Aldehydes (11a-13a-d)
entry |
aldehyde |
Conditions product |
yield % a ( E/Z ratio) b |
1 |
13a |
1 N NaOH, r.t. 1 N NaOH, 90 oC |
65 (56:44) |
3 |
13b |
1 N NaOH, r.t. 1 N NaOH, 90 0C |
80 (46:54) |
5 |
13c |
1 N NaOH, r.t. | 95 (48:52) |
6 |
11a |
1N NaOH, r.t. 1 N NaOH, 90 0C |
38 (49:51) |
8 |
13d |
1 N NaOH, r.t. | 63 (54:46) |
a Yields of isolated stilbenes ( 14a-e). b Measured by 1H NMR of crude mixture.
The isolated yields of the alkenes 14a-e by this method were good to excellent, proving that the rate of reaction of the ylide with the substrate aldehydes is indeed much faster than decomposition to 9. Isolation of the stilbenes ( 14a-e) was achieved by partitioning the reaction mixture with CH2Cl2, followed by removal of the water by addition of anhydrous magnesium sulfate, and then precipitation of the polymer supported triphenylphosphine oxide byproduct 9 into diethyl ether and concentration of the etheric solution to dryness.To explore the scope of this reaction two components were varied: base strength and temperature. Increasing the base-strength (from 1 N to 2 N NaOH) neither affected the yield of stilbenes ( 14a-e) nor the E:Z isomer distribution. However, elevating the temperature (up to 90 0C) resulted in improved yields of 14d and as to be expected an increase in the E:Z ratio for the stilbene products.
1.4. Regeneration of Spent Reagent 9. A number of standard methods were employed to regenerate the oxidized polymer-supported phosphine 9 (Table 3). Only poor to moderate reduction of 9 to 2 occurred with polymethylhydrosiloxane (PMHS)35 using various conditions. The reason is perhaps due to unfavorable polymer-polymer-interactions between the polymeric reductant and the PEG-backbone. Trichlorosilane,36,37 while leading to a significant reduction of the phosphine group (56 %), completely cleaved the terminal phosphine from the polymer. However, quantitative reduction of 9 was achieved with freshly prepared alane (AlH 3)38 in THF as determined by 31P NMR vide supra. The polymer was isolated in 75 % yield following the usual precipitation into degassed ether. The incomplete polymer recovery is attributed to problems associated with the byproduct aluminium salts either complexing to the polymer support and thus preventing precipitation or polymer-backbone hydrolysis during the reduction process. Thus, the utility of this reagent was made complete by its facile regeneration by quantitative reduction and reisolation.
Table 3. Regeneration of PEG-supported triarylphosphine 2.
entry |
reagent |
conditions |
P(V) to P(III) a/ % conversion |
1 |
PMHSb |
CH2Cl2/ r.t. |
20 |
2 |
PMHS |
Benzene / reflux 20 h |
33 |
3 |
PMHS |
THF / reflux 20 h |
40 |
4 |
HSiCl3c |
Benzene / reflux 20 h |
56 |
5 |
AlH3THF |
THF / r.t. 30 min |
100 |
aMeasured by 31P NMR of the reaction mixtures. bPolymethylhydrosiloxane.cWith triethylamine.
The Stille39,40 cross-coupling reaction possesses wide scope and functional group tolerance. In addition, the requisite tin reagents are easily prepared and relatively stable when compared to most organometallic reagents. These properties make this particular process very attractive as a route to many carbon-carbon bond forming reactions under relatively ambient conditions. The importance of this reaction within the sphere of polymer-supported chemistry has been demonstrated with the recent emergence of solid-41-43 and fluorous-phase44,45 approaches.The defining feature of the Stille coupling is the use of a trialkyltin reagent in a palladium-catalyzed coupling with either a halide or triflate. As with all tin chemistry, the benefits associated with these reagents are to some extent outweighed by the problems associated with their removal after reaction completion. The tri- n-butyl derivatives are more difficult to remove from a reaction mixture than the methyl congeners due to their low volatility and poor water solubility. Therefore there is an increasing need for methodologies which facilitate the separation of the side-product tributyltin derivatives from the required adducts following the Stille reaction. Our aim was to study the potential of poly(ethylene glycol) as a soluble matrix for the electrophile component of the reaction. The tributyltin derivatives and other components of the reaction being in solution could, theoretically, be separated from the polymer-supported products by a simple precipitation of the support into a suitable solvent and recovery by filtration.MeO-PEG5000 was initially esterified with either para- or ortho-iodobenzoic acid using DCC and DMAP to give para- and ortho-polymer-supported iodides, 15a-b respectively, with polymer recoveries being > 95 %.46 The conversion was quantitative based upon 1H NMR spectroscopic analysis of 15a-b.A number of efficient reaction conditions for solution-phase Stille couplings have been developed,39,40,47,48 and initially we utilized a number of these for the reaction between PEG-iodide 15a and tributyl phenyltin ( 16) to explore the viability of PEG as a soluble polymer support for this reaction and then to identify the optimal conditions necessary for the PEG-supported Stille variant (Scheme 3 and Table 4). Previous work has revealed conflicting evidence regarding the potential for coordination of the PEG polyether backbone to transition metals.49,50 Initially therefore, we had serious concerns that the PEG-backbone may serve to either retard or completely inhibit the Stille reaction via complexation of either the palladium catalyst, the alkali metal halide or the organostannane reagent.
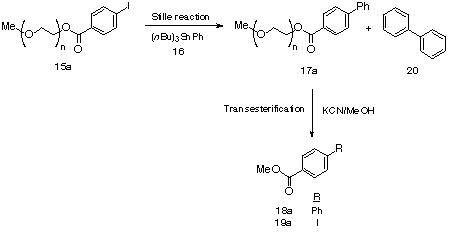
Table 4. Optimization of the Stille cross-coupling reaction parameters between 15a and 16
entry |
catalyst |
solvent |
conc. / mMa |
T / oC |
LiCl |
time / h |
yield 18a / % b |
yield 19a / % b |
1 |
Pd(PPh3)2Cl2 |
toluene |
20 |
110 |
+ |
24 |
22 |
-c |
2 |
Pd(PPh3)2Cl2 |
THF |
20 |
reflux |
+ |
24 |
68 |
-c |
3 |
Pd(PPh3)2Cl2 |
DMF |
20 |
80 |
+ |
24 |
89 |
-c |
4 |
Pd(PPh3)4 |
toluene |
20 |
110 |
+ |
24 |
53 |
-c |
5 |
Pd(PPh3)4 |
THF |
20 |
reflux |
+ |
24 |
7 |
73 |
6 |
Pd(PPh3)4 |
DMF |
20 |
80 |
+ |
24 |
10 |
58 |
7 |
Pd(PPh3)2Cl2 |
DMF |
20 |
80 |
- |
24 |
86 |
12 |
8 |
Pd(PPh3)2Cl2 |
DMF |
20 |
80 |
+ |
48 |
97 |
0 |
9 |
Pd(PPh3)2Cl2 |
DMF |
10 |
80 |
+ |
24 |
98 |
0 |
a Based on the concentration of PEG-bound substrate 15a. b Isolated yields after workup.c Not determined.
The efficiency of the initial series of experiments was determined both by 1H NMR analysis of the PEG-biaryl adduct 17a and, following transesterification, by the isolated yields of the biaryl methyl ester 18a and monoaryl ester 19a (resulting from no cross-coupling with 15a). Reactions were conducted in toluene, THF or DMF, with either a Pd(0) or Pd(II) catalyst (0.1 equiv.) in the presence or absence of LiCl (10 equiv.). A three-fold excess of tributyl phenylstannane 16 was used relative to the PEG-supported electrophile 15a. Additional parameters that were modified included temperature, reaction time and reaction concentration. solvent (Table 1, entries 3 and 6-9) under standard conditions of 15a (20 mM) and 80 0C. The results show that DMF is the most superior of the solvents tested for the PEG-supported Stille reaction. Similar to the case with THF, PdCl2(PPh3)2 is the best catalyst (Table 4, entries 3 and 6) with considerable amounts of the transesterified iodide 19a being recovered after reaction with Pd(0) (58 %). In addition, by leaving the reaction to run for 48 h rather than the preliminary 24 h, a considerable increase in the observed biaryl adduct 18a is achieved (89 and 97 % respectively) (Table 4, entries 3 and 8).In a recent fluorous-phase approach to the Stille reaction, between aryl tin reagents and aryl halides, it was observed that by using LiCl as an additive that the cross-coupling efficiencies were significantly enhanced.44 However, the positive effect of LiCl on our PEG-supported Stille variant seems to be only marginal (compare Table 4, entries 3 and 7). This result is more in line with previous reports which found that LiCl does not usually enhance the reaction between trialkyl tin reagents with aryl halides, but is generally more useful with aryl triflate electrophiles.51,52The summary of these results is that the use of PdCl2(PPh3)2 in the presence of LiCl in DMF at a reaction concentration of 20 mM for 48 h, or 10 mM for 24 h, gives excellent yields for the monomethoxy-PEG 5000-supported variant of the Stille cross-coupling reaction (97 and 98 % respectively).This liquid-phase approach imparts a number of significant advantages over its solution-phase counterpart. The toxic tributyl tin derivatives are easily separated from the PEG-biaryl adduct 17a by precipitation of the polymer-support into either iso-propyl alcohol or diethyl ether. Polymer recovery was >99 % in each case. In addition, the homocoupled biaryl side-product 20, an incontrovertible product in the solution-phase reaction,39,40 does not contaminate the soluble-polymer supported biaryl product 17a, because the homocoupling side-reaction takes place in solution and thus the contaminant is removed during the precipitation step. Having optimized the liquid-phase Stille reaction conditions on monomethoxy-PEG5000, we then sought to examine the scope of this cross coupling process in a parallel format. A range of tributyl stannanes, 16 and 21-28, were reacted with the PEG-supported iodides 15a-b in a parallel fashion, furnishing the library of biaryl, heterobiaryl and styryl derivatives 18a-b and 28a-b to 34a-b (Table 5).

The isolated yields of all library members were good to excellent (69 - 99 %) showing that under our pre-optimized conditions vide supra, that the scope of the PEG-supported variant of the Stille reaction is quite broad. Following passage down a short pad of silica gel, the library members ( 18a-b, 28a-b to 34a-b ) were each isolated in > 95 % purity.In each case (Table 5, entries 1-8) the yield of the ortho-isomer was lower than its corresponding para-congener. We rationalize that this phenomenon may be a result, at least in part, of the steric effect imparted by the PEG-backbone polymer chain during ligand transfer. However this effect followed no definitive trend related to the size of the moiety being transferred. suggests that the process involved may be more complex than simple steric retardation. While, in this report, no attempt has been made to re-optimize the reaction conditions to improve the yield of the ortho-isomers, it is speculated that future studies directed to resolve this issue would involve simply increasing the number of equivalents of the stannane and/or palladium catalyst, whilst perhaps using more dilute reaction conditions.
Acknowledgments. Financial support for this work was supplied by the NIH (GM 56154, KDJ) and The Skaggs Institute for Chemical Biology (KDJ).
References and Notes
1. Shuttleworth, S. J.; Allin, S. M.; Sharma, P. K. Synthesis 1997,
27, 1217-1239.
2. Sherrington, D. C.; Hodge, P. In Synthesis and Separations Using
Functional Polymers ,John Wiley & Sons, Inc.: New York, 1988.
3. Gravert, D. J.; Janda, K. D. Chem. Rev. 1997, 97,
489-509.
4. Geckeler, K. E. Adv. Polym. Sci. 1995, 121, 31-79.
5. Wentworth Jr., P.; Janda, K. D. Curr. Opin. Biotech. 1998,
9, 109-115.
6. Booth, R. J.; Hodges, J. C. Acc. Chem. Res. 1999, 32,
18-26.
7. Han, H.; Wolfe, M. M.; Brenner, S.; Janda, K. D. Proc. Natl. Acad.
Sci. USA 1995, 92, 6419-6423.
8. Han, H.; Janda, K. D. J. Am. Chem. Soc. 1996, 118,
7632-7633.
9. Sieber, F.; Wentworth Jr., P.; Toker, J. D.; Wentworth, A. D.; Metz, W.
A.; Reed, N. N.; Janda, K. D. J. Org. Chem. 1999, 64, 5188-5192.
10. Wentworth Jr., P.; Vandersteen, A. M.; Janda, K. D. Chem. Commun. 1997,
759-760.
11. Hori, M.; Janda, K. D. J. Org. Chem. 1998, 63,
889-894.
12. Zhao, X.-Y.; Janda, K. D. Tetrahedron Lett. 1997, 38,
5437-5440.
13. Zhao, X.-Y.; Janda, K. D. Bioorg. & Med. Chem. Lett. 1998,
8, 2439-2442.
14. Chen, S.; Janda, K. D. J. Am. Chem. Soc. 1997, 119,
8724-8725.
15. Chen, S.; Janda, K. D. Tetrahedron Lett. 1998, 39,
3943-3946.
16. Wentworth Jr., P.; Vandersteen, A. M.; Janda, K. D. Chem. Commun. 1997,
759-760.
17. Wipf, P.; Venkatraman, S. Tetrahedron Lett. 1996, 37,
4659-4663.
18. Harris, J. H.; Liu, Y.; Chai, S.; Andrews, M. D.; Vederas, J. C. J.
Org. Chem. 1998, 63, 2407-2409.
19. A recent report attempts the synthesis of 2 for use as a
macroinitiator in block copolymer synthesis (Choi, Y. K.; Bae, Y. H.; Kim, S. W. Macromolecules
1995, 28, 8419-8421). However, the authors observed significant oxidation of
the phosphine end-groups during its isolation which they did not isolate in pure form.
20. A recent report descibes a one step route to phosphine 6 in
moderate yield (63 % yield).[Herd, H.; Heȼler, A.; Hingst, W.; Tepper, M.; Stelzer, O. Journal
of Organometallic Chemistry 1996, 522, 69-76.] However, in our hands the
obtained yields were even lower (38%). Therefore we adopted a three step approach to 6
the overall yield being 83 % (Scheme 1).
21. Zhao, X.-Y.; Janda, K. D. Tetrahedron Lett. 1997, 38,
5437-5440.
22. Relles, H. M.; Schluenz, R. W. J. Am. Chem. Soc. 1974, 96,
6469-6475.
23. Yan, B. Acc. Chem. Res. 1998, 31, 621-630.
24. Feiser, M.; Feiser, L. F. In Reagents for Organic Synthesis Mir,
Moscow: USSR, 1978 pp 1238-1239.
25. Ferraboshi, P.; Gambero, C.; Azadani, M. N.; Santaniello, E. Synth.
Commun. 1986, 16, 667-672.
26. Li, C.-J.; Chan, T.-H. In Organic Reactions in Aqueous Media ,
John Wiley & Sons, Inc.: New York, 1997.
27. Li, C.-J. Chem. Rev. 1993, 93, 2023-2035.
28. Lubineau, A.; Augȩ, J.; Queneau, Y. Synthesis 1994,
741-760.
29. McKinley, S. V.; Rakshys, J. W. J. Chem. Soc. Chem. Commun. 1972,
134-135.
30. Heitz, W.; Michels, R. Angew. Chem. Int. Ed. Engl. 1972,
11, 298-299.
31. Heitz, W.; Michels, R. Justus Liebigs Ann. Chem. 1973,
227-230.
32. Clarke, S. D.; Harrison, C. R.; Hodge, P. Tetrahedron Lett. 1980,
21, 1375-1378.
33. Hodge, P.; Waterhouse, J. Polymer 1981, 22,
1153-1154.
34. Bernard, M.; Ford, W. T. J. Org. Chem. 1983, 48,
326-332.
35. Lipowitz, J.; Bowman, S. A. J. Org. Chem. 1973, 38,
162-165.
36. Horner, L.; Balzer, W. D. Tetrahedron Lett. 1965, 3,
1157-1158.
37. Cremer, S. E.; Chorvat, R. J. J. Org. Chem. 1967, 32,
4066-4070.
38. Griffin, S.; Heath, L.; Wyatt, P. Tetrahedron Lett. 1998,
39, 4406.
39. Stille, J. K. Pure & Appl. Chem. 1985, 57,
1771-1780.
40. Stille, J. K. Angew. Chem. Int. Ed. Engl. 1986, 25,
508-524.
41. Deshpande, M. S. Tetrahedron Lett. 1994, 35,
5613-5614.
42. Forman, F. W.; Sucholeiki, I. J. Org. Chem. 1995, 60,
523-528.
43. Plunkett, M. J.; Ellman, J. A. J. Am. Chem. Soc. 1995, 117,
3306-3307.
44. Hoshino, M.; Degenkolb, P.; Curran, D. P. J. Org. Chem. 1997,
62, 8341-8349.
45. Curran, D. P.; Hoshino, M. J. Org. Chem. 1996, 61,
6480-6481.
46. Blettner, C. G.; König, W. A.; Stenzel, W.; Schotten, T. Synlett
1998, 295-297.
47. Mitchell, T. N. Synthesis 1992, 803-815.
48. Farina, V. Pure & Appl. Chem. 1996, 68,
73-78.
49. Bergbreiter, D. E.; Liu, Y.-S. Tetrahedron Lett. 1997, 38,
3703-3706.
50. Blettner, C. G.; König, W. A.; Stenzel, W.; Schotten, T. Synlett
1998, 803-815.
51. Scott, W. J.; Stille, J. K. J. Am. Chem. Soc. 1986, 108,
3033-3037.
52. Chen, Q.-Y.; He, Y.-B. Chin. J. Chem. 1990.
All comments on this poster should be sent by e-mail to (mailto:[email protected] ona.edu)
[email protected] with B0007 as the message subject of your e-mail.