Fourth International
Electronic Conference on Synthetic Organic Chemistry (ECSOC-4),
www.mdpi.org/ecsoc-4.htm, September 1-30, 2000
[C0028]
Synthesis of some anilides of substituted pyrazine-2-carboxylic acids
and their photosynthesis-inhibiting activity
Martin Dolezal1a*,
Miroslav Miletin1,
Jiri Hartl1,
Katarina Kralova2,
Jiri Kunes3
1Department of Pharmaceutical Chemistry and Drug Control, Faculty of Pharmacy, Charles University, 500 05 Hradec Kralove, Czech Republic
aE-mail: [email protected],
Tel.: +420 49 5067272, Fax: +420 49 5512423
2Institute of Chemistry, Faculty of Natural Sciences, Comenius University,
842 15 Bratislava, Slovak Republic
3Department of Inorganic and Organic Chemistry, Faculty of Pharmacy,
Charles University, 500 05 Hradec Kralove, Czech Republic
* Author to whom correspondence should be addressed.
Received: 9 August 2000 /
Uploaded: 14 August
Abstract: Condensation of chlorides of substituted pyrazine-2-carboxylic acids with ring substituted anilines yielded a series of anilides of 6-chloropyrazine-2-carboxylic, 5-(1,1-dimethylethyl)-pyrazine-2-carboxylic or 6-chloro-5-(1,1-dimethylethyl)pyrazine-2-carboxylic acids. Products were tested for their photosynthesis-inhibiting activity. The most active inhibitor of oxygen evolution rate in spinach chloroplasts was 3,5-bis(trifluoromethyl)anilide of 6-chloropyrazine-2-carboxylic acid (IC50 = 0.026 mmol dm-3), the most active antialgal compound was 3-methylanilide of 6-chloro-5-(1,1dimethylethyl)pyrazine-2-carboxylic acid (IC50 = 0.063 mmol dm-3). The elimination of phenolic group in the benzene moiety led to an decreased photosynthesis-inhibitory activity.
Keywords: Anilides of 6-chloro-5-alkylpyrazine-2-carboxylic acids; Photosynthesis inhibition; Spinach chloroplasts; Chlorella vulgaris.
Introduction
Various compounds possessing -NHCO- group, e. g. acyl and thioacyl anilides, benzanilides, phenyl carbamates, etc., were found to inhibit photosynthetic electron transport [1--4]. Anilides of 2-alkylpyridine-4-carboxylic [5,6], 2-alkylsulfanyl-4-pyridinecarboxylic [6,7] acids inhibited oxygen evolution rate in Chlorella vulgaris and their inhibitory activity depended on the lipophilicity of the compounds. Several esters of alkoxy substituted phenylcarbamic acids (APA) showed the antialgal activity against Chlorella vulgaris [8--10]. The inhibitory efficiency of APA concerning chlorophyll production in Chlorella vulgaris depended on the lipophilicity of the alkoxy substituent and also on its position on the aromatic ring [8--10]. The antialgal activity of APA correlated with the antifungal activity of these compounds against Candida albicans [10]. We have recently reported the synthesis of a series of anilides prepared from some or 5-alkylpyrazine-2-carboxylic acids and some aminophenols [11].
The presented study is concerned in the synthesis of another series of anilides prepared from substituted pyrazin-2-carboxylic acids and alkylated, resp. alkoxylated (2-, 3-methyl; 2,6-dimethyl, 2-methoxy) or halogenated (3-bromo; 3,5-bis(trifluoromethyl)) anilines. The aim of this work is to search for the structure--activity relationships and to determine the importance of increased hydrophobic properties for photosynthesis-inhibiting evaluation of newly prepared pyrazine-2-carboxylic acid anilides.
Results and Discussion
The synthesis of anilides is shown in Scheme 1. Condensation of chlorides of 6-chloropyrazine-2-carboxylic (1a)[12], 5-(1,1-dimethylethyl)pyrazine-2-carboxylic (1b) [11] or 6-chloro-5-(1,1-dimethylethyl)pyrazine-2-carboxylic (1c) [11] acids with ring substituted anilines yielded a series of 18 anilides of mentioned substituted pyrazine-2-carboxylic acids 2a--r.
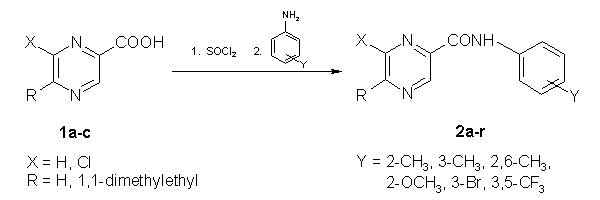
Scheme 1: Preparation of anilides of substituted pyrazine-2-carboxylic acids 2a--r.
The melting points, yields, and elemental analyses for the compounds prepared are given in Table 1, and the IR and 1H NMR spectral data in Table 2. Calculated log P values of anilides 2a--r are shown in Table 3.
Biological activity of prepared anilides 2a--r concerning inhibition of oxygen evolution rate in spinach chloroplasts was investigated. The inhibitory activity of the compounds has been expressed as IC50 values (see Table 3). Additionally some inhibition of chlorophyll production in green algae Chlorella vulgaris was studied at the anilides 2f, 2l, 2m, 2n, 2o and 2p. Results of their antialgal activity are given in Table 4.
The studied compounds inhibited photosynthetic electron transport in spinach chloroplasts what was reflected in the inhibition of oxygen evolution rate. The IC50 values varied in the range from 0.026 (2m) to 1.072 mmol dm-3 (2a). In general, the photosynthesis-inhibiting activity of the studied compounds depended on their lipophilicity showing a quasi-parabolic course. However, the studied compounds could be divided into two group. The compounds with 2-CH3 substituent on the phenyl ring (2a, 2b, 2c, 2p, 2q and 2r) had lower biological activity than the other investigated compounds with comparable log P values. Consequently, we assume that the methyl substituent in ortho position of the benzene ring is disadvantageous from the viewpoint of interactions with the photosynhetic apparatus. On the other hand, compound 2m exhibited higher inhibitory activity than expected.
The antialgal activity of six studied compounds (2f, 2l, 2m, 2n, 2o and 2p) showed a quasi-parabolic dependence upon log P with maximum activity for compounds having log P in the range from 3.18 to 5.16. With the further increasing of the lipophilicity a dramatic decrease of antialgal activity was observed.
Table 1. Analytical data of the anilides 2a--r.
| Compd. |
X |
R |
Y |
Formula
M. w. |
% Calculated
% Found |
M. p. / oC
Yield / % |
| C |
H |
N |
F |
Cl |
Br |
| 2a |
Cl |
H |
2-CH3 |
C12H10ClN3O
247.7 |
58.19
58.02 |
4.07
4.14 |
16.97
16.86 |
-- |
14.31
14.19 |
-- |
97--99
75 |
| 2b |
H |
(CH3)3C |
2-CH3 |
C16H19N3O
269,3 |
71.35
71.48 |
7.11
7.08 |
15.60
15.67 |
-- |
-- |
-- |
80--81
84 |
| 2c |
Cl |
(CH3)3C |
2-CH3 |
C16H18ClN3O
303.8 |
63.26
63.15 |
5.97
5.82 |
13.83
13.96 |
-- |
11.67
11.86 |
-- |
114--115
78 |
| 2d |
Cl |
H |
3-CH3 |
C12H10ClN3O
247.7 |
58.19
58.08 |
4.07
4.11 |
16.97
16.80 |
-- |
14.31
14.48 |
-- |
83--84
79 |
| 2e |
H |
(CH3)3C |
3-CH3 |
C16H19N3O
269,3 |
71.35
71.41 |
7.11
7.22 |
15.60
15.77 |
-- |
-- |
-- |
94--95
85 |
| 2f |
Cl |
(CH3)3C |
3-CH3 |
C16H18ClN3O
303.8 |
63.26
63.40 |
5.97
6.08 |
13.83
14.01 |
-- |
11.67
11.74 |
-- |
98--99
84 |
| 2g |
Cl |
H |
2-OCH3 |
C12H10ClN3O2
263.7 |
54.66
54.57 |
3.82
3.93 |
15.94
16.01 |
-- |
13.45
13.35 |
-- |
71--72
85 |
| 2h |
H |
(CH3)3C |
2-OCH3 |
C16H19N3O2
285.3 |
67.35
67.16 |
6.71
6.68 |
14.73
14.62 |
-- |
-- |
-- |
77--78
88 |
| 2i |
Cl |
(CH3)3C |
2-OCH3 |
C16H18ClN3O2
319.8 |
60.09
60.16 |
5.67
5.59 |
13.14
13.23 |
-- |
11.09
11.07 |
-- |
118--119
82 |
| 2j |
Cl |
H |
3-Br |
C11H7BrClN3O
312.5 |
42.27
42.37 |
2.26
2.25 |
13.44
13.41 |
-- |
11.34
11.48 |
25.56
25.60 |
99--100
83 |
| 2k |
H |
(CH3)3C |
3-Br |
C15H16BrN3O
334.2 |
53.91
54.03 |
4.83
4.97 |
12.57
12.61 |
-- |
-- |
23.91
23.77 |
113--114
75 |
| 2l |
Cl |
(CH3)3C |
3-Br |
C15H15BrClN3O
368.7 |
48.87
48.79 |
4.10
4.22 |
11.40
11.28 |
-- |
9.62
9.77 |
21.67
21.78 |
104--105
62 |
| 2m |
Cl |
H |
3,5-CF3 |
C13H6ClF6N3O
369.7 |
42.24
42.21 |
1.64
1.66 |
11.37
11.33 |
30.84
30.77 |
9.59
9.46 |
-- |
132--133
88 |
| 2n |
H |
(CH3)3C |
3,5-CF3 |
C17H15F6N3O
391.3 |
52.18
52.02 |
3.86
3.84 |
10.74
10.72 |
29.13
29.17 |
-- |
-- |
135--137
89 |
| 2o |
Cl |
(CH3)3C |
3,5-CF3 |
C17H214ClF6N3O
425.8 |
47.96
48.01 |
3.31
3.41 |
9.87
9.63 |
26.77
26.56 |
8.33
8.51 |
-- |
98--99
88 |
| 2p |
Cl |
H |
2,6-CH3 |
C13H12ClN3O
361.7 |
59.66
59.70 |
4.62
4.70 |
16.06
16.09 |
-- |
13.55
13.67 |
-- |
121--122
75 |
| 2q |
H |
(CH3)3C |
2,6-CH3 |
C18H22ClN3O
331.8 |
72.06
72.09 |
7.47
7.45 |
14.83
14.84 |
-- |
-- |
-- |
84--85
78 |
| 2r |
Cl |
(CH3)3C |
2,6-CH3 |
C18H22ClN3O
331.8 |
64.25
64.19 |
6.34
6.40 |
13.22
13.18 |
-- |
11.16
11.17 |
-- |
145--146
68 |
Table 2. IR and 1H NMR spectral data of the anilides 2a--r.
| Compd. |
IR (cm-1)
(C=O) |
1H-NMR (delta, ppm; J in Hz) |
| NH |
H3 |
H5 |
H6 |
H2' |
H3' |
H4' |
H5' |
H6' |
C(CH3)3 |
OCH3 |
CH3 |
| 2a |
1692 |
9.42bs |
9.40s |
8.81s |
-- |
-- |
7.33--7.22m |
7.33--7.22m |
7.17--7.10m |
8.15--8.11m |
-- |
-- |
2.40s |
| 2b |
1685 |
9.71bs |
9.41dd
J=1.37 |
8.65dd
J=1.37 |
-- |
-- |
7.33--7.20m |
7.33--7.20m |
7.10td
J=7.70 |
8.26d
J=7.70 |
1.45s |
-- |
2.40s |
| 2c |
1695 |
9.42s |
9.28s |
-- |
-- |
-- |
7.32--7.21m |
7.32--7.21m |
7.12td
J=7.41 J=1.37 |
8.18--8.13m |
1.56s |
-- |
2.40s |
| 2d |
1692 |
9.35bs |
9.39bs |
8.80s |
-- |
7.61--7.52m |
-- |
7.61--7.52m |
7.28t
J=7.96 |
7.03--6.98m |
-- |
-- |
2.39s |
| 2e |
1684 |
9.61s |
9.40d
J=1.51 |
-- |
8.62d
J=1.51 |
7.65--7.61m |
-- |
7.56--7.50m |
7.27t
J=7.69 |
6.98d
J=7.69 |
1.45s |
-- |
2.38s |
| 2f |
1694 |
9.32bs |
9.27s |
-- |
-- |
7.63--7.59m |
-- |
7.57--7.51m |
7.28t
J=7.69 |
7.02--6.97m |
1.55s |
-- |
2.39s |
| 2g |
1690 |
10.04s |
9.38s |
8.78s |
-- |
-- |
6.94dd
J=7.96 J=1.64 |
7.13td
J=7.69 J=1.51 |
7.03td
J=7.69 J=1.51 |
8.52dd
J=7.96 J=1.64 |
-- |
3.97s |
3.97s |
| 2h |
1691 |
10.27bs |
9.39d
J=1.37 |
-- |
8.68d
J=1.37 |
-- |
6.94dd
J=7.96 J=1.64 |
7.11td
J=7.69 J=1.53 |
7.02td
J=7.69 J=1.53 |
8.59dd
J=7.96 J=1.64 |
1.45s |
3.96s |
-- |
| 2i |
1695 |
10.01bs |
9.26s |
-- |
-- |
-- |
6.94dd
J=7.97 J=1.51 |
7.12td
J=7.97 J=1.51 |
7.02td
J=7.97 J=1.51 |
8.53dd
J=7.97 J=1.51 |
1.55s |
3.97s |
-- |
| 2j |
1701 |
9.38bs |
9.38bs |
8.82s |
-- |
8.01t
J=1.92 |
-- |
7.67ddd
J=7.96 J=1.92
J=1.37 |
7.35--7.22m |
7.35--7.22m |
-- |
-- |
-- |
| 2k |
1692 |
9.66bs |
9.38d
J=1.65 |
-- |
8.62d
J=1.65 |
8.03t
J=1.92 |
-- |
7.66dt
J=7.65 J=1.92 |
7.32--7.21m |
7.32--7.21m |
1.45s |
-- |
-- |
| 2l |
1697 |
9.36bs |
9.26s
|
-- |
-- |
8.02t
J=1.92 |
-- |
7.66dt
J=7.69 J=1.92 |
7.34--7.22m |
7.34--7.22m |
1.55s |
-- |
-- |
| 2m |
1681 |
9.66bs |
9.41s |
8.87s |
-- |
8.87bs |
-- |
7.70bs |
-- |
8.87bs |
-- |
-- |
-- |
| 2n |
1699 |
9.94bs |
9.41d
J=1.51 |
-- |
8.64d
J=1.51 |
8.28bs |
-- |
7.66bs |
-- |
8.28bs |
1.46s |
-- |
-- |
| 2o |
1686 |
9.63bs |
9.29s |
-- |
-- |
8.29bs |
-- |
7.68bs |
-- |
8.29bs |
1.56s |
-- |
-- |
| 2p |
1691 |
8.94bs |
9.39s |
8.83s |
-- |
-- |
7.21--7.10m |
7.21--7.10m |
7.21--7.10m |
-- |
-- |
-- |
2.28s |
| 2q |
1667 |
9.16bs |
9.40d
J=1.37 |
-- |
8.65d
J=1.37 |
-- |
7.19--7.09m |
7.19--7.09m |
7.19--7.09m |
-- |
1.46s |
-- |
2.29s |
| 2r |
1710 |
8.91bs |
9.27s |
-- |
-- |
-- |
7.21--7.07m |
7.21--7.07m |
7.21--7.07m |
-- |
1.57s |
-- |
2.28s |
Table 3. IC50 values concerning inhibition of oxygen evolution rate in spinach chloroplasts by the tested anilides 2a--r and calculated log P values of the compounds in comparison with standard (atrazine).
| Compd. |
IC50[mmol dm-3] |
log P |
| 2a |
1.072 |
2.72 +- 0.41 |
| 2b |
0.440 |
3.28 +- 0.40 |
| 2c |
0.244 |
4.41 +- 0.42 |
| 2d |
0.486 |
2.72 +- 0.41 |
| 2e |
0.148 |
3.28 +- 0.40 |
| 2f |
0.118 |
4.41 +- 0.42 |
| 2g |
--a |
2.15 +- 0.42 |
| 2h |
0.286 |
2.72 +- 0.41 |
| 2i |
0.097 |
3.84 +- 0.43 |
| 2j |
0.313 |
3.46 +- 0.48 |
| 2k |
0.081 |
4.03 +- 0.48 |
| 2l |
0.107 |
5.15 +- 0.50 |
| 2m |
0.026 |
5.16 +- 0.54 |
| 2n |
0.114 |
5.73 +- 0.53 |
| 2o |
0.241 |
6.85 +- 0.55 |
| 2p |
0.649 |
3.18 +- 0.41 |
| 2q |
0.229 |
3.75 +- 0.40 |
| 2r |
0.242 |
4.87 +- 0.42 |
| Atrazine |
0.001 |
1.03 +- 0.62 |
| a not measured (insoluble compound) |
Table 4. IC50 values concerning inhibition of chlorophyll production in green algae Chlorella vulgaris by the tested anilides 2f, 2l, 2m, 2n, 2o and 2p and calculated log P values of the compounds.
| Compd. |
IC50[mmol dm-3] |
log P |
| 2f |
0.063 |
4.41 +- 0.42 |
| 2l |
0.067 |
5.15 +- 0.50 |
| 2m |
0.125 |
5.16 +- 0.54 |
| 2n |
0.208 |
5.73 +- 0.53 |
| 2o |
0.356 |
6.85 +- 0.55 |
| 2p |
0.079 |
3.18 +- 0.41 |
Experimental
General
Melting points were determined on a Kofler apparatus and are uncorrected. Elemental analyses were obtained using an EA 1110 CE instrument (Fisons Instruments S. p. A., Milan). The IR spectra were recorded on a Nicolet Impact 400 spectrometer in KBr pellets. The 1H NMR spectra were measured for solutions in CDCl3 with a Varian Mercury - Vx BB 300 spectrometer operating at 300 MHz. Chemical shifts were recorded as delta values in parts per million (ppm), and were indirectly referenced to tetramethylsilane via the solvent signal (7.26 for 1H). Multiplicities are given together with the coupling constants (in Hz). Log P values were computed using a program ACD/Log P ver. 1.0 (Advanced Chemistry Development Inc., Toronto).
Synthesis of anilides 2a--r
A mixture of acid (i. e. 6-chloropyrazine-2-carboxylic [8], 5-(1,1-dimethylethyl)pyrazine-2-carboxylic [3] or 6-chloro-5-(1,1-dimethylethyl)-pyrazine-2-carboxylic [3] acids, 0.05 mol) and thionyl chloride (5.5 cm3, 75 mmol) in 20 cm3 of dry benzene was refluxed for about 1 h. Excess of thionyl chloride was removed by repeated evaporation with dry benzene in vacuo. The crude acyl chloride dissolved in 50 cm3 of dry acetone was added dropwise to a stirred solution of the corresponding substituted aniline (50 mmol) in 50 cm3 of dry pyridine keeping at the room temperature. After the addition was complete, stirring continued for another 30 min. The reaction mixture was then poured into 200 cm3 of cold water and the crude anilide was collected and recrystallized from aqueous ethanol.
Study of inhibition of oxygen evolution rate in spinach chloroplasts
The oxygen evolution rate in spinach chloroplasts was investigated spectrophotometrically (Specord UV VIS, Zeiss, Jena) in the presence of an electron acceptor 2,6-dichlorophenol--indophenol, by method described in Ref. [13]. The compounds were dissolved in dimethyl sulfoxide (DMSO) because of their low water solubility. The used DMSO volume fractions (up to 5 vol. %) did not affect the oxygen evolution. The inhibitory efficiency of the studied compounds has been expressed by IC50 values, i. e. by molar concentration of the compounds causing 50 % decrease in the oxygen evolution relative to the untreated control. Comparable IC50 value for a selective herbicide atrazine [14] is about 1.0 mmol dm-3.
Study of inhibition of chlorophyll production in green algae Chlorella vulgaris
The algae Chlorella vulgaris were cultivated statically at room temperature according to Sidoova et al. [15] (photoperiod 16 h light / 8 h dark; illumination 4000 lx; pH = 7.2). The effect of compounds 2f, 2l, 2m, 2n, 2o and 2p on algal chlorophyll (Chl) content was determined after 4-day cultivation in the presence of the tested compounds, expressing the response as percentage of the corresponding values obtained for control. The Chl content in the algal suspension was determined spectrophotometrically (Specord UV VIS, Zeiss Jena, Germany) after extraction into N,N-dimethylformamide according to Inskeep and Bloom [16]. The Chl content in the suspensions at the beginning of cultivation was 0.5 mg dm-3. Because of their low water solubility, the tested compounds were dissolved in DMSO. DMSO concentration in the algal suspensions did not exceed 0.25 v/v % and the control samples contained the same DMSO amount as the suspensions treated with the tested compounds.
Acknowledgements. This study was supported by the Grant Agency of Charles University in Prague (Grant No. 26/1998 BCH), by the Scientific Intents of Charles University in Prague (No. 11160001), by the Ministry of Education of the Czech Republic (project No. VS97124), and by the Scientific Grant Agency of the Ministry of Education of the Slovak Republic and the Slovak Academy of Sciences (Grant No. 1/7262/20). We also thank D. Karlickova, J. Zizkova, and T. Vojtisek for their skillful technical assistance and Dr. D. Mikulasova from the Department of Biochemistry, Faculty of Natural Sciences, Comenius University, Bratislava, for her assistance in the preparation of chloroplasts.
References
[1] Good N. E.: Plant. Physiol. 1961, 36, 788.
[2] Kralova K., Sersen F., Cizmarik J.: Chem. Pap. 1992, 46, 266.
[3] Kralova K., Sersen F., Miletin M., Hartl J.: Chem. Pap. 1998, 52, 52.
[4] Kralova K., Sersen F., Kubicova L., Waisser K.: Chem. Pap. 1999, 53, 328.
[5] Miletin M., Hartl J., Machacek M.: Collect. Czech. Chem. Commun. 1997, 62, 672.
[6] Kralova K., Loos D., Miletin M., Klimesova V.: Folia Pharm. Univ. Carol. 1998, 23 Suppl., 77.
[7] Miletin M., Dolezal M., Hartl J., Kralova K., Machacek M.: International Electronic Conference on Synthetic Organic Chemistry (ECSOC-3), September 1--30, 1999, c0005.
[8] Kralova K., Loos D., Cizmarik J.: Collect. Czech. Chem. Commun. 1994, 59, 2293.
[9] Kralova K., Loos D., Cizmarik J.: Photosynthetica 1994, 30, 155.
[10] Kralova K., Bujdakova H., Cizmarik J.: Pharmazie 1995, 50, 440.
[11] Dolezal M., Hartl J., Miletin M., Machacek M., Kralova K.: Chem. Pap. 1999, 53, 126.
[12] Abe Y., Shigeta Y., Uchimaru F., Okada S., Ozasayama E.: Japan. 69 12,898 (1969); Chem. Abstr. 1969, 71, 112979y.
[13] Kralova K., Sersen F., Sidoova E.: Chem. Pap. 1992, 46, 348.
[14] Carpentier R., Fuerst E. P., Nakatani H. Y., Arntzen C. J.: Biochim. Biophys. Acta 1985, 808, 293.
[15] Sidoova E., Kralova K., Mitterhauszerova L.: Chem. Pap. 1992, 46, 55.
[16] Inskeep W. P., Bloom P. R.: Plant Physiol. 1985, 77, 483.
All comments on this poster should be sent by e-mail to (mailto:[email protected]
ona.edu)
[email protected]
with C0028 as the message subject of your e-mail.

