Oxygen-Containing
10-, 15-, and 20-Membered Macrocyclic Cobalt Complexes
from
Co2(CO)6-Bispropargylic
Alcohol
David Díaz Díaz 1,*,†
Juan Pedro Ceñal 2 and Víctor S. Martín 1,*
1 Instituto
Universitario de Bio-Orgánica “Antonio González”, Universidad de La
Laguna, Avda. Astrofísico Francisco Sánchez, 2, 38206 La Laguna,
Tenerife, Spain. Tel: (+34)-922-318579; Fax: (+34)-922-318571.
2 INTEQUI-CONICET-Facultad de
Química, Bioquímica y Farmacia, Universidad Nacional de San Luis,
Chacabuco y Pedernera, 5700-San Luis, Argentina.
† Present
Address: The Dow Chemical Company (Dow Europe GmbH), Bachtobelstrasse
3, CH 8810 Horgen, Switzerland.
* Author to whom
correspondence should be addressed; E-mail: [email protected]
Received: 6 November
2007; in revised form: 22 January 2008
/ Accepted: 23 January 2008 / Published: 25 March 2008
Abstract:
Novel 10-, 15-, and 20-membered
oxygen-containing macrocyclic
cobalt complexes (3, 4,
and 5 respectively) were obtained
in moderate combined yield via double
nucleophilic substitution of the hexacarbonyldicobalt-coordinated
monoyne diol
[{Co2(CO)6(μ-η2-HOCH2C≡CCH2OH)}]
(1) upon BF3·OEt2
treatment in the presence of (Z)-2-butene-1,4-diol
(2) at room temperature. The products distribution was
found to be highly
concentration dependent.
Keywords:
Nicholas
reaction, cobalt, alkyne complexes, macrocycles
The
Nicholas reaction
is a powerful and versatile synthetic tool in organic synthesis. It
basically
consists in the nucleophilic attack on a Co2(CO)6-propargylic
cation, which is generated by adding either protic acids or Lewis
acids, mainly
trifluoromethane sulfonic acid, HBF4·OEt2
and BF3·OEt2 on Co2(CO)6-propargylic
alcohols. The reaction works well with a variety of nucleophiles to
form new
bonds between the propargylic carbon and atoms that include carbon,
oxygen,
hydrogen and nitrogen. The procedure can be applied inter- or
intra-molecularly
in solution or in solid phase [1].
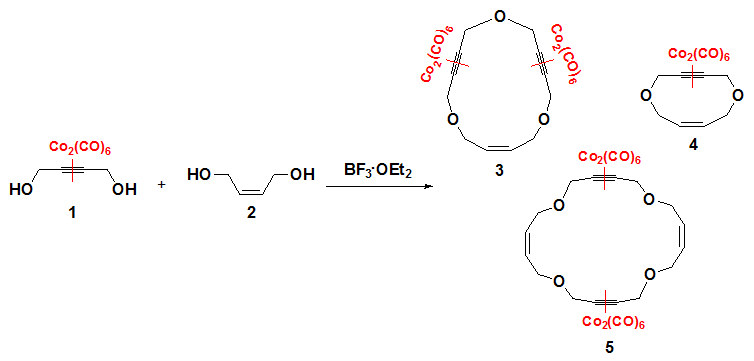
The treatment
of the
hexacarbonyldicobalt-coordinated
monoyne diol [{Co2(CO)6(μ-η2-HOCH2C≡CCH2OH)}]
(1) [2] with two
equivalents of BF3·OEt2
and one equivalent of (Z)-2-buten-1,4-diol
(2) at
room temperature under diluted conditions afforded a mixture of
the macrocycles 3, 4, and 5 in
62% combined yield. The ratio of the isolated macrocycles was
found to be highly dependent on the concentration conditions. Thus,
under 0.01M
dilution, 4 was obtained
as the major product (see
Experimental Section), whereas 5 was the
major one under higher molar concentration (0.02M). The
oxygen-containing macrocycles [3] were separated by silica gel
column chromatography
and their molecular weights confirmed by mass spectroscopy [4].
Experimental Section
General
1H
and 13C NMR spectra were
recorded at 25 °C on Bruker Avance-300 spectrometer in CDCl3
as
solvent, and chemical shifts are reported relative to Me4Si.
Low-
and high-resolution mass spectra were obtained by using a Micromass
Autospec
spectrometer. Elemental analysis was performed on a Fisons Instrument
EA 1108
CHNS-O analyzer. Infrared spectra were recorded on a Bruker IFS 55
spectrophotometer
on compounds dispersed on a CaF2 disc
(20 × 2 mm). Column
chromatographies were performed on Merck silica gel, 60 Å and 0.2-0.5
mm. Methylene chloride was dried
by distillation over calcium
hydride prior to use. All reagents
were commercially available and used without further
purification. Hexacarbonyldicobalt-coordinated
monoyne diol [{Co2(CO)6(μ-η2-HOCH2C≡CCH2OH)}]
(1) was prepared as reported in the
literature [2].
Synthesis of cobalt-complexed oxygen-containing
macrocyclics 3-5:
To a stirred solution of [{Co2(CO)6(μ-η2-HOCH2C≡CCH2OH)}]
(1) (200 mg, 0.54 mmol) in CH2Cl2
(54 mL, 0.01M) were consecutively added (Z)-2-butene-1,4-diol
(2) (45
mL,
0.54 mmol) and BF3·OEt2
(137 ml, 1.08 mmol) under an
argon atmosphere at room temperature. The reaction mixture was
monitored by
TLC. The mixture was poured with
vigorous stirring into
a saturated solution of NaHCO3 at 0 °C for 15
min and extracted with
CH2Cl2. The combined organic phases
were washed with brine, dried (MgSO4),
concentrated, and the
products separated by silica gel column chromatography.
Macrocycles 3, 4, and 5
were isolated as red oils in
a ratio 1:10:4 respectively and 62% combined yield. Interestingly,
under more
concentrated conditions (0.02M) the ratio of the macrocycles was found
to be
1:3:8 respectively. It is worth to mention that traces of other cobalt
complexes were also formed along with the isolated
macrocycles,
although these minor
products could not be isolated and characterized in the preliminary
studies.
8,13-Bis-(hexacarbonyldicobalt)-μ2-{η2-1,6,11-trioxa-cyclopentadec-3-ene-8,13-diyne}
(3):
1H NMR (300 MHz, CDCl3)
d = 4.28 (m, 4H), 4.71 (s, 4H), 4.95
(s, 4H), 5.76 (m,
2H); 13C NMR (75 MHz, CDCl3)
d = 66.6 (t), 71.0 (t), 130.6 (d),
199.2 (s); IR
(thin film) υ (cm-1)
2093, 2053, 2022; FAB-MS m/z (relative
intensity) 779 (M+1)+ (8) , 778 (M)+ (30), 722 (M−2CO)+ (12), 694 (M−3CO)+ (10), 638 (M−5CO)+ (13), 610 (M−6CO)+ (17), 553 (100). HMRS
calcd for C24H14Co4O15
(M-1)+
777.76606, found 777.76680.
3-Hexacarbonyldicobalt-μ2-{η2-3,4-didehydro-2,5,7,10-tetrahydro-1,6-dioxecino}
(4):
1H NMR (300 MHz, CDCl3)
d = 4.23 (m, 4H), 4.71 (s, 4H), 5.76
(m, 2H); 13C
NMR (75 MHz, CDCl3) d =
66.5 (t), 70.6 (t), 129.3 (d), 194.3 (s); IR (thin film) υ (cm-1) 2094, 2054, 2022; FAB-MS m/z
(relative intensity) 425 (M+1)+ (22) , 424 (M−1)+ (18), 396 (M−CO)+ (22), 368 (M−2CO)+ (38), 365 (100), 340
(M−3CO)+ (16), 312 (M−4CO)+ (29). HMRS calcd
for C14H9Co2O8
(M-1)+
422.89614, found 444.89578.
8,18-Bis-(hexacarbonyldicobalt)-μ2-{η2-1,6,11,16-tetraoxa-cycloicosa-3,13-diene-8,18-diyne}
(5):
1H
NMR (300 MHz, CDCl3) d = 4.45 (m, 8H),
4.88 (s, 8H), 5.86
(m, 4H); 13C NMR (75 MHz, CDCl3)
d = 66.6
(t), 71.0 (t), 130.6 (d); IR (thin film) υ (cm-1)
2060, 2032, 2012; FAB-MS m/z (relative
intensity) 849 (M+1)+ (26) , 848 (M)+ (21), 820 (M−CO)+ (20), 764
(M−3CO)+ (33), 736
(M−4CO)+ (30), 708
(M−5CO)+ (29), 680
(M−6CO)+ (21), 652
(M−7CO)+ (21), 624
(M−8CO)+ (26), 602
(100), 596 (M−9CO)+ (20). Anal.
Calcd for
C28H20Co4O16: C, 39.65; H,
2.38. Found: C,
39.86; H, 2.51.
Acknowledgements
This research was
financially supported by the
Ministerio de Educación y Ciencia of Spain,
co-financed by the European
Regional Development Fund (CTQ2005-09074-C02-01/BQU) and the Canary
Islands
Government.
References
and Notes
- For
reviews on
the chemistry uses of cobalt-complexed propargylic cations and related
issues,
see: (a) Nicholas, K. M. Acc. Chem. Res. 1987, 20,
207-221; (b) Caffyn,
A. J. M.; Nicholas, K. M. In Comprehensive Organometallic
Chemistry II;
Hegedus, L. S., Ed.; Pergamon: Oxford, 1995; Vol. 12, Chapter 7.1, pp
685-702; (c)
Welker, M. E. Current
Organic Chemistry 2001, 5, 785-807; (d) Green, J. R. Current Organic Chemistry 2001,
5, 809-826; (e) Müller, T. J. J. Eur. J. Org. Chem. 2001,
2021-2033; (f) Teobald, B. J. Tetrahedron
2002, 58,
4133-4170; (g) Fryatt, R.; Christie S. D. R. J.
Chem. Soc., Perkin Trans. 1 2002,
447-4
- For other uses of
1 as masked
dielectrophiles in cyclization reactions, see: (a)
Gruselle,
M.;
Malézieux, B.; Vaissermann, J.;
Amouri, H. Organometallics
1998, 17,
2337-2343; (b) Lu, Y.; Green, J. R. Synlett 2001,
243-247; (c) Soleilhavoup, M.; Saccavini,
C.; Lepetit, C.; Lavigne,
G.; Maurette, L.; Donnadieu, B.; Chauvin, R. Organometallics
2002, 21,
871-883.
- For a complete study on the
synthesis of oxygen-containing
macrocycles from the analog 1,3-diyne diol [{Co2(CO)6(μ-η2-HOCH2C2-)}2],
see: Hope-Weeks, L. J.; Mays, M. J.; Solan, G. A. Eur.
J. Inorg. Chem. 2007,
3101-3114.
- For the synthesis
of sulfur-containing macrocycles from 1,
see: (a)
Gelling, A.; Jeffery, J. C.; Povey, D. C.; Went, M. J. J.
Chem. Soc., Chem. Commun. 1991,
349-351; (b)
Dernirhan, F.; Irişli,
S.; Salek, S. N.; Şentük, O. S.; Went, M. J.; Jeferry, J. C. J. Organomet. Chem. 1993,
453, C30-C31; (c) Dernirhan, F.; Gelling, A.;
Irişli, S.; Jeferry, J. C.;
Salek, S. N.; Şentük, O. S.; Went, M. J. J.
Chem. Soc. Dalton Trans. 1993,
2765-2773;
(d) Bennett,
S. C.; Jeffery, J.
C.; Went, M. J. J. Chem. Soc. Dalton
Trans. 1994, 3171-3176; (e) Davies, J.
E.; Hope-Weeks, L.
J., Mays, M. S.; Raithby, P. R. Chem.
Commun. 2000, 1411-1412.
©
2008 by MDPI (http://www.mdpi.org/).
Reproduction is permitted for noncommercial purposes.
