Molbank
2008, M571
![]()
molbank
ISSN 1422-8599
www.mdpi.org/molbank
Short Note
7-Hydroxy-8-acetylcoumarin N-1-(Carboxymethyl)pyridinium
Chloride Hydrazone
Antigoni
Kotali 1,*, Ioannis S. Lafazanis 1 and Philip A. Harris
2
1 Laboratory of Organic Chemistry,
Department of Chemical Engineering, College of Engineering, University of
Thessaloniki, Thessaloniki 54124, Greece E-mail: [email protected]
2 GlaxoSmithKline, 1250 South Collegeville Road, P.O.Box
5089, Collegeville, PA 19426-0989, USA E-mail: [email protected]
*
Author to whom correspondence should be addressed.
Received:
24 April 2007; in revised form: 8 August 2008 /
Accepted: 18 August 2008 / Published: 24
August 2008
Keywords: 7-hydroxy-8-acetylcoumarin, carbonylhydrazones.
As part of a research programme
targeting novel molecules with possible biological activity, derived from o-hydroxyaryl
ketone hydrazones [1] we attempted to synthesize a water soluble hydrazone of 7-hydroxy-8-acetylcoumarin. Thus, we
used 1-(carboxymethyl)pyridinium chloride
hydrazide (Girard’s Reagent P), a well known reagent that
produces water soluble ketone derivatives.
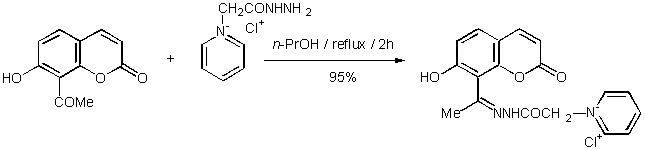
7-Hydroxy-8-acetylcoumarin
was prepared according to the literature method [2] whereas, commercially
available Girard’s Reagent P was supplied by Aldrich. Girard’s
Reagent P (0.92 g, 4.9 mmol) was added to a solution of 7-hydroxy-8-acetylcoumarin (1 g, 4.9 mmol) in 1-propanol (25
mL). The
reaction mixture was refluxed for 2 hours. It was then allowed to cool at room
temperature. Subsequently, it was stored in the refrigerator overnight. Filtration
of the precipitate, which was formed, afforded of the desired hydrazone as white crystals (1.73
g, 95 %). The product was identified by its 1H NMR, 13C NMR and MS without
further purification and was found to be a mixture of two isomers as shown by 1H and 13C NMR data.
M.p. 248-249 °C.
1H NMR (400 MHz, DMSO-d6):
2.22 (s, 3H), 2.37 (s, 3H), 5.79 (s, 2H), 5.82 (s, 2H), 6.16-6.22 (m, 2H),
6.98-7.11 (m, 2H), 7.50-7.56 (m, 2H), 7.91-7.97 (m, 2H), 8.13-8.19 (m, 4H),
8.61-8.66 (m, 2H), 9.02-9.11 (m, 4H), 10.60 (s, 1H), 10.70 (s, 1H), 11.18 (s,
1H), 11.46 (s, 1H).
13C NMR (100 MHz, DMSO-d6):
19.1, 20.0, 62.0, 62.4, 109.7, 111.8, 111.9, 112.0, 112.4, 113.7, 113.8, 113.9,
114.4, 115.5, 128.1, 128.2, 128.3, 129.9, 130.1, 130.3, 145.6, 145.9, 147.0,
147.1, 147.9, 152.9, 153.2, 153.3, 158.8, 159.7, 160.0, 160.8, 161.0, 162.9,
167.1, 167.9.
MS m/z
(ESI+) : Calcd. for C18H16N3O4Cl: 711.19647
[C36H32N6O8Cl]+,
675.21979 [C36H31N6O8]+,
338.11353 [C18H16N3O4]+.
Found: 711.19687 [C36H32N6O8Cl]+,
675.22022 [C36H31N6O8]+,
338.11320 [C18H16N3O4]+.
References
|
1. |
Kotali, A.; Harris, P. A. Org. Prep.
Proc. Int. 1994, 26(2), 155. |
|
|
2. |
Abramov, M. A.; Dehaen, W. Synthesis, 2000, 11, 1529. |
|
© 2008 by the authors; licensee Molecular
Diversity Preservation International, Basel, Switzerland. This article is an
open-access article distributed under the terms and conditions of the Creative
Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).