Molbank 2008, M575
![]()
molbank
ISSN 1422-8599
www.mdpi.org/molbank
Short Note
Synthesis of 5,17,29- tri[(4-methyl-1-piperazinyl)methyl]-11,23,35-tri-tert-butyl-37,39,41-trimethoxy-calix[6]arene
Rachid Souane* and Jacques Vicens
Laboratoire
de Conception Moléculaire, IPHC-ECPM-UMR 7512 (CNRS-ULP), 25 rue Becquerel,
F-67087 Strasbourg Cedex 2, France
* Author to whom correspondence should be addressed. E-mail: [email protected]; Tel. +33-3-90242695
Received: 23 July 2008; in revised form: 2 September 2008 / Accepted: 4 September 2008 / Published: 4 September 2008
Keywords: Calixarene, piperazine.
1. Introduction
A calixarene is a macrocycle or
cyclic oligomer based on a condensation product of a
phenol and an aldehyde [1]. The word calixarene is derived from calix
or chalice because this type of molecule resembles a vase and from the word arene that refers to the aromatic building blocks. Calixarenes have hydrophobic cavities that can hold smaller
molecules or ions and belong to the class of cavitands
known in Host-Guest chemistry. Calixarene nomenclature is straightforward and involves counting the number of
repeating units in the ring and include it in the name. A calix[4]arene has four units in the ring and a calix[6]arene has six.
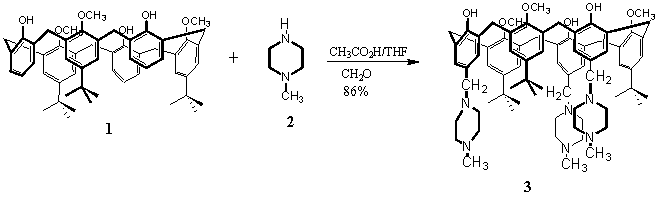
2. Experimental
CH2O (0.189 g, 6.372 mmol) and 1-methyl piperazine (2) (0.370 g, 4.25 mmol) was added to solution of tri-tert-butyl trimethoxy calix[6]arene (1) [2] (0.300 g, 0.35 mmol) in THF (10 ml) and acetic acid (10 ml). The mixture was stirred for 64 h at room temperature. The solvents were removed under reduced pressure and water (20 ml) was added. Afterwards, a 10% solution of K2CO3 in water (40ml) was added. The product 3 precipitated (0.242 g, 86 %).
Mp 158-160 °C.
1H NMR (200 MHz, CDCl3): δH 6.99 (s, 6H+3H, ArH+ArOH); 6.89 (s, 6H, ArH); 3.88 (s, 12H, ArCH2Ar); 3.54 (s, 9H, ArOCH3); 3.38 (s, 6H, ArCH2N); 2.43 (s large, 24H, NCH2CH2NCH2CH2); 2.27 (s, 9H, NCH3), 1.03 (s, 27H, C(CH3)3).
Anal Found: C, 75.89; H, 8.31%. Calc. for C75H102O6N6: C, 76.10; H, 8.69%.
FAB(+)MS:
m/z = 1183.8 (M)+, 1182.8 (M–H)+.
References
1. Gutsche,
C.D. Calixarenes; Royal Society of
Chemistry:
2. Casnati, A.; Domiano, L.; Pochini, A.; Ungaro, R.; Carramolino, M.; Oriol-Magrans, J.; Nieto, P.M.; Lopez-Prados, J.; Prados, P.; de Mendoza, J.; Janssen, R. G.; Verboom, W.; Reinhoudt, D.N. Tetrahedron 1995, 51, 12699-12720.
© 2008
by the authors; licensee Molecular Diversity Preservation International,